Originally published May 4, 2026, as a BioBoyScout white paper. Republished here on Substack with full content, embedded charts, and downloadable PDF. — Robert
A Deep-Dive Conviction Analysis of ARO-MAPT’s Phase 1/2a Readout Across Three Lines of Evidence, Twelve Primate Brain Regions, and a 50-70 Percent CSF Tau Reduction Range
Robert Toczycki, JD, MBA
bioboyscout@gmail.com
847.227.7909
X: @BioBoyScout
Arrowhead Pharmaceuticals will read out human Phase 1/2a data for ARO-MAPT, its subcutaneous tau-lowering drug for Alzheimer’s disease, in the second half of 2026. The case for a position before that readout rests on three converging lines of preclinical and clinical evidence: a tau target already clinically validated by BIIB080, a subcutaneous transferrin-receptor (TfR1) delivery route structurally better matched to the deep brain regions where Alzheimer’s pathology starts, and primate data predicting a 50 to 70 percent cerebrospinal fluid tau reduction in humans. This is corroborated by Arrowhead’s revealed-preference behavior across partnership structure, CNS portfolio expansion, and manufacturing capacity options. The argument is not that ARO-MAPT cannot fail, the argument is that the structure of evidence assembled to date justifies meaningful conviction in the readout, well above the generic Phase 1 base rate.
Companion to The Endgame, the fourth paper in the BioBoyScout white paper series
Executive Summary
Arrowhead Pharmaceuticals will read out human Phase 1/2a data for ARO-MAPT, its subcutaneous tau-lowering drug for Alzheimer’s disease, in the second half of 2026. The case for entering a position before that readout rests on three converging lines of preclinical and clinical evidence, assembled across two and a half years of progressively detailed scientific disclosure, corroborated by Arrowhead’s own revealed-preference behavior.
The tau target is de-risked. BIIB080, a competitor drug developed by Biogen and Ionis using intrathecal delivery, has demonstrated in a 46-patient Phase 1b trial that lowering tau in Alzheimer’s patients produces 63 to 67 percent reductions in cerebrospinal fluid tau, reductions in aggregated tau pathology across all six brain regions scanned, and directional cognitive trends matching the approved amyloid antibodies. The biology has been validated by a competitor. ARO-MAPT inherits that validation.
The delivery route is structurally superior to the established alternative. Subcutaneous injection through Arrowhead’s transferrin-receptor-mediated platform distributes drug uniformly across brain tissue. Intrathecal injection produces a surface-to-deep gradient with weakest concentrations in the deep medial temporal regions where Alzheimer’s pathology starts. Arrowhead’s primate data, presented at the December 2025 CNS Delivery Summit, shows 70 to 80 percent tau messenger RNA reduction across all twelve brain regions tested, including the deep regions intrathecal struggles to reach.
A successful Phase 1/2a is sufficient as a strategic catalyst. Subcutaneous delivery of RNA interference (RNAi) drugs to the brain has been the largest unsolved problem in the modality for two decades. Arrowhead is the company that built the platform that solves it. ARO-MAPT is the first drug on the platform to enter the clinic, with dosing under way as of December 2025. SRP-1005 (formerly ARO-HTT, licensed to Sarepta) is the second clinical program, with first-in-human dosing planned for the second quarter of 2026. ARO-SNCA (licensed to Novartis at the preclinical stage) is the third. As of April 2026, ARO-MAPT remains the only clinical-stage subcutaneous RNAi CNS program in development; the most recent independent preclinical disclosure of the same mechanistic configuration (Dyne Therapeutics, April 27, 2026) is years from human dosing and is being positioned for partnership rather than internal advancement. The 2026 ARO-MAPT readout would be the first human clinical validation of the platform. Once that validation arrives, every potential pharmaceutical acquirer faces both the offensive question (how much is the platform worth to acquire) and the defensive question (how much is it worth to keep it from a competitor). Novartis and Sarepta have already committed substantial diligence-stage capital against the platform; the price of clinical validation will sit on top of those commitments. Section IV develops the platform argument and partnership economics in detail.
The case is corroborated by Arrowhead’s revealed-preference behavior. The company retained ARO-MAPT and the broadest tauopathy indications while monetizing the periphery through Novartis and Sarepta partnerships, has staked seven CNS programs across the platform before any human validation, and holds a 6.24-acre land option adjacent to its Verona manufacturing facility that expires in 2028, the binding decision on which sits in management’s hands roughly eighteen months after the readout. Each is a forward commitment of capital or commercial intent that only makes sense if leadership believes the platform will be validated.
The case is not without risk. The primate-to-human translation could fail. The cognitive benefit suggested by BIIB080’s directional Phase 1b data may not replicate at scale. The Phase 1/2a sample size is small enough that a noisy result is possible. The history of failed anti-tau antibody programs creates a default skepticism that is reasonable. The Bottom Line section addresses each of these risks in detail. The honest framing is that the case justifies meaningful conviction, not certainty.
I. The Question
A. Why This Paper Exists Now
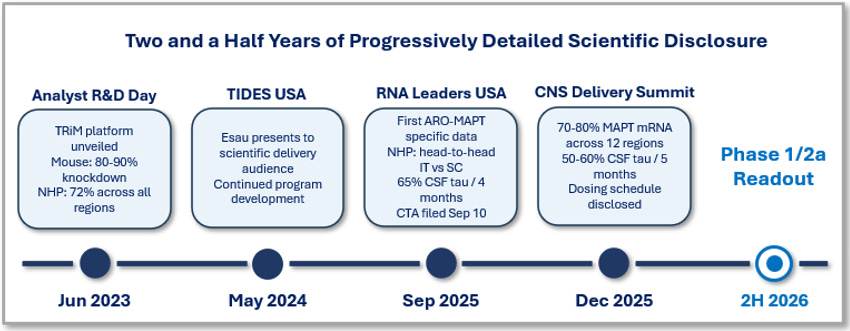
Arrowhead Pharmaceuticals has been disclosing scientific progress on its central nervous system program at increasing levels of detail for two and a half years. The company has presented at four separate venues, with each disclosure adding meaningful new evidence over the prior one. The first came at the June 2023 Analyst R&D Day, where Arrowhead introduced its blood-brain-barrier-crossing TRiM platform with preliminary mouse data showing 80 to 90 percent knockdown via subcutaneous administration and primate data showing 72 percent knockdown across all brain regions, including the deep brain. The second came at TIDES USA in May 2024, where Christine Esau, the company’s Vice President of Biology, presented the same platform to a scientific delivery-focused audience, confirming continued internal commitment to the subcutaneous CNS approach. The third came at the RNA Leaders USA Congress on September 10, 2025, where Esau presented the first ARO-MAPT-specific dataset: a head-to-head primate comparison showing subcutaneous administration produces uniform deep-brain knockdown while intrathecal does not, MAPT messenger RNA reductions across twelve primate brain regions, and a 65 percent cerebrospinal fluid tau reduction sustained after four months of repeat dosing in non-human primates (Source: Esau, RNA Leaders USA Congress, September 10, 2025, slide 20). On the same day, Arrowhead disclosed that it had filed the Clinical Trial Application in New Zealand for the human Phase 1/2a study. The fourth, and most detailed to date, came at the 7th Annual CNS Delivery Summit on December 10, 2025, where Agnieszka Glebocka, Ph.D., the company’s Vice President of Discovery Chemistry, presented the full scientific package: 70 to 80 percent MAPT messenger RNA reduction across all twelve brain regions tested, 50 to 60 percent cerebrospinal fluid tau reduction sustained for five months, and the dosing schedule for the human program.
Figure 1. Two and a half years of progressively detailed scientific disclosure on Arrowhead’s subcutaneous CNS platform, from the June 2023 Analyst R&D Day (mouse and preliminary primate data) through the December 2025 CNS Delivery Summit (full ARO-MAPT preclinical package). The 2H 2026 Phase 1/2a readout is the next disclosure milestone.
Across these four disclosures, the underlying data has been internally consistent and progressively more rigorous. The story Arrowhead is telling in December 2025 is the same story it began telling in June 2023, but with more detailed scientific evidence at each step and with ARO-MAPT-specific data emerging in September 2025. This pattern is meaningful. It suggests the company has been confident enough in its central nervous system platform to keep disclosing publicly, in increasing detail, for two and a half years. It also suggests that the December 2025 data should not be interpreted as a one-off slide deck. It is the latest milestone in a steady, publicly visible scientific program that began with mouse data in 2023 and has now produced primate data sufficient to support a human clinical trial.
The Endgame, the fourth paper in the BioBoyScout white paper series, asked what Arrowhead Pharmaceuticals would be worth if ARO-MAPT succeeds. The answer ran 84 pages: a 12-asset inventory, strategic value analysis methodology, competitive bidder landscape, fully diluted per-share acquisition price math. The valuation work was done. What was not done was the harder question that every reader of The Endgame eventually asked: how confident should we be that ARO-MAPT will succeed in the first place?
That is the question this paper answers. The Setup is not a valuation paper. The Endgame already did that work, and that work stands on its own. This paper takes the valuation case as given and assembles the evidence that supports the underlying clinical event: a strong cerebrospinal fluid tau readout in the second half of 2026. The reader’s job is to weigh the evidence and arrive at their own level of conviction. The paper’s job is to lay out the evidence as completely and as honestly as the available data allows.
B. What This Paper Is Not
Three things this paper is not. First, it is not a valuation paper. The Endgame did that work, and readers who want the per-share acquisition math should read that paper. This paper is about the underlying clinical event. Second, it is not a forecast of what ARWR stock will do in the second half of 2026. The realized stock price will depend on the actual readout, on the market’s reaction, and on broader conditions outside the scope of this paper. Third, this is not a paper that argues ARO-MAPT cannot fail. It can. The Bottom Line section acknowledges the residual risks, including primate-to-human translation, the small sample size of the Phase 1/2a, and the historical track record of anti-tau drugs in Alzheimer’s disease. Each risk is real. The argument is not that failure is impossible, the argument is that the structure of evidence assembled to date justifies a meaningful level of conviction in the readout, well above the generic Phase 1 base rate.
With those caveats made explicit, the paper assembles its case across the rest of the sections. The paper begins with the strongest argument, that the underlying tau biology has already been clinically validated by a competitor, and builds from there. By the time the reader reaches the synthesis sections, the argument should land naturally. The reader can then apply their own judgment to the strength of the evidence as a whole.
The 2026 readout will produce its own answer. This paper is about what the appropriate level of conviction should be when that answer arrives, and what the structure of the evidence already assembled tells us about how to weigh that answer.
The Endgame asked what ARWR would be worth if ARO-MAPT succeeds. The Setup asks how confident we should be that ARO-MAPT will succeed. The scientific answer is built on four publicly disclosed scientific presentations spanning two and a half years, on independent peer-reviewed primate work characterizing how each delivery route performs in the brain, on the clinically validated tau biology that a competitor has already demonstrated, and on the structural anatomy of how drugs distribute through the brain. That scientific case is corroborated by Arrowhead’s own revealed-preference behavior in partnership structure, CNS portfolio expansion, and manufacturing capacity options. The readout in the second half of 2026 will produce the verdict. This paper is about how to read it when it arrives.
II. The Tau Target Is De-Risked
A. The Bottom Line First
Someone else’s drug already proved that tau works as a target in Alzheimer’s disease. That drug is BIIB080, developed by Biogen and Ionis, and its Phase 1b trial in 46 mild Alzheimer’s patients produced the strongest body of clinical evidence any tau-targeting therapy has ever generated. By the end of the long-term extension, BIIB080 had reduced cerebrospinal fluid tau by 63 to 67 percent at the high doses, reduced aggregated tau pathology across all six tau positron emission tomography regions of interest scanned, and produced cognitive trends in the same range as the approved amyloid antibodies.
This matters for ARO-MAPT for one specific reason. The hardest question any new drug has to answer is whether the underlying biology actually works, whether changing the target produces the disease benefit the developer claims it will. BIIB080 has answered that question. The biology works. Tau lowering produces measurable, sustained biomarker effects in real patients with real Alzheimer’s disease.
ARO-MAPT does not have to discover that tau works. BIIB080 already did the discovery. ARO-MAPT only has to demonstrate that its specific molecule, delivered through subcutaneous injection rather than spinal injection, can match what BIIB080 achieved. That is a fundamentally easier question to answer than the one most Phase 1/2a candidates face. Most Phase 1/2a candidates are simultaneously asking whether the target works and whether their molecule works. ARO-MAPT is only asking the second question.
This section walks through what BIIB080 actually showed, what those results mean for ARO-MAPT, and why the question of clinical success in 2026 is structurally different from a generic Phase 1/2a coin flip.
B. What “Target De-Risked” Actually Means
Drug development asks two separate questions. First: does changing this molecular target actually help the patient? This is the target validation question, and it is a question about biology. Second: can this particular molecule, with its specific chemistry and delivery profile, change the target effectively and safely? This is the compound validation question, and it is a question about a specific drug.
These two questions fail in different ways. Drugs fail target validation when the underlying science turns out to be wrong, the disease isn’t actually caused by what we thought it was caused by. Drugs fail compound validation when the science is right but the specific molecule has problems: it can’t reach the target tissue, it has off-target effects, it can’t be dosed at therapeutically relevant levels, it triggers an immune response.
BIIB080 separated these two questions for ARO-MAPT. Because BIIB080 already demonstrated that lowering tau produces measurable disease-relevant effects, the target validation question for ARO-MAPT is largely answered before the trial begins. The Phase 1/2a in 2026 is testing the compound validation question for ARO-MAPT specifically. That is a meaningful and answerable question, but it is not the same question as testing whether the underlying biology works.
C. What BIIB080 Actually Showed
BIIB080 (also called MAPTRx) is a spinal-injection drug developed by Biogen and Ionis. Technically it is an antisense oligonucleotide (ASO), a synthetic strand designed to bind and silence the messenger RNA that codes for tau protein. Mechanistically, BIIB080 works through different molecular machinery than ARO-MAPT (Section III explains the difference), but both drugs target the same underlying biology: lower tau messenger RNA, lower tau protein, lower tau pathology in the brain.
The BIIB080 Phase 1b trial enrolled 46 patients with confirmed mild Alzheimer’s disease across 12 centers in Canada, Finland, Germany, the Netherlands, Sweden, and the United Kingdom. Patients received the drug by spinal injection, with the highest doses administered every three months. The trial ran for an initial 36-week placebo-controlled phase (13 weeks of dosing followed by a 23-week post-dosing follow-up), and patients could then enter a long-term extension where every patient eventually received the active drug. The headline results, published in Nature Medicine in 2023 and presented at a major Alzheimer’s conference later that year, are striking.
Three results matter for the ARO-MAPT thesis. First, BIIB080 produced large, sustained, dose-dependent reductions in cerebrospinal fluid tau, in the 63 to 67 percent range at the high doses, after roughly two years of follow-up. This is not a marginal effect. It is one of the largest pharmacodynamic effects any central nervous system drug has demonstrated in mild Alzheimer’s disease, comparable in pharmacological terms to what the approved ALS drug tofersen achieves. Second, BIIB080 reduced aggregated tau pathology measured by tau positron emission tomography. The reductions were present across all six prespecified regions of interest, including the deep medial temporal lobe where Alzheimer’s pathology starts. Tau positron emission tomography is a regional measurement, not whole-brain, so the precise framing is that all six imaged regions showed reductions, not that every neuron in the brain was reached.
Third, BIIB080 produced directional cognitive benefits suggestive of clinical activity. When the Phase 1b participants were compared against a matched control group drawn from the contemporaneous TANGO trial, the high-dose patients showed approximately 2.0 to 2.4 points of advantage on the Clinical Dementia Rating Sum of Boxes scale at week 100, against an estimated TANGO placebo decline of approximately 3.1 points. Two important caveats apply. The Phase 1b sample of 46 patients was too small for those differences to reach statistical significance. The TANGO comparator group was not randomized to BIIB080, so the comparison is hypothesis-generating rather than benefit-quantifying, the kind of analysis that motivates a properly powered Phase 2 trial rather than substituting for one. With those caveats acknowledged, the direction was consistent across every clinical scale measured, and the magnitude, if reproduced under randomized conditions, would substantially exceed what the approved amyloid antibodies achieved, lecanemab at 27 percent slowing on the same scale and donanemab at 36 percent. The properly powered test of whether tau lowering produces statistically significant cognitive benefit is the BIIB080 Phase 2 (CELIA) study, which reads out in mid-2026.
D. Why This Matters for ARO-MAPT
The BIIB080 dataset establishes three things that flow directly into the ARO-MAPT investment thesis.
First, tau lowering is achievable in real Alzheimer’s patients. This is no longer a hypothesis. There is a published clinical trial showing it in 46 humans. ARO-MAPT does not need to discover this. ARO-MAPT only needs to demonstrate that its molecule, delivered through its route, achieves comparable effects in its trial.
Second, tau lowering translates into measurable downstream effects. The cerebrospinal fluid tau reduction was not an isolated biomarker. It was accompanied by reductions in phosphorylated tau (the form most directly linked to Alzheimer’s pathology), by reductions in aggregated tau across the brain regions where the disease actually lives, and by directional cognitive benefits that pattern-match the approved amyloid antibodies. The chain from messenger RNA reduction to cognitive effect is no longer hypothetical. It has been observed.
Third, the bar for ARO-MAPT’s Phase 1/2a is now specific rather than abstract. The Phase 1/2a does not have to ask whether tau lowering produces effects. It only has to ask whether ARO-MAPT, delivered subcutaneously, produces cerebrospinal fluid tau reductions broadly comparable to BIIB080’s 63 to 67 percent. The ARO-MAPT primate data, characterized in Section III.E, predicts cerebrospinal fluid tau reductions in the 50 to 70 percent range in humans, which would either match BIIB080 or fall modestly below it while still representing a pharmacologically meaningful effect. That is a specific, measurable bar.
Two studies published in early 2026 reinforce the case for upstream protein-pathology intervention as the right therapeutic strategy in Alzheimer’s disease. A UC Irvine team (Nakagawa et al., Nature Neuroscience 2026) showed in an APP-knock-in mouse model that amyloid pathology drives downstream dopamine dysfunction in the entorhinal cortex, the same deep memory region where Alzheimer’s pathology starts. The dopamine deficit is a consequence of upstream protein pathology, not an independent disease mechanism. The therapeutic implication is that addressing the upstream protein buildup matters more than treating downstream neurochemical consequences in isolation. ARO-MAPT works at the upstream level by reducing the production of one of those toxic proteins. A UC San Diego team (Jati et al., Acta Neuropathologica Communications 2026) identified a natural protective mechanism in patients who carry full amyloid and tau pathology yet remain cognitively intact, the so-called asymptomatic Alzheimer’s disease state. Roughly twenty to thirty percent of older individuals fall into this category. Patients who progress to clinical dementia are the ones whose natural protection has failed, and for them the link between toxic protein buildup and cognitive decline holds. Those are the patients ARO-MAPT will treat. A companion paper from the same UC San Diego group (Jati et al., bioRxiv 2026) is itself characterizing a therapeutic intervention to lower tau pathology through an adrenergic-inhibition mechanism distinct from RNAi, which underscores that the field treats tau pathology as a real and consequential drug target. The 2026 literature continues to validate the underlying biology that BIIB080 first demonstrated clinically.
In drug development, there are two ways a drug can fail. The first is that the target is wrong, the disease isn’t actually caused by what scientists thought it was caused by. The second is that the target is right but the specific drug doesn’t work, it can’t reach the right tissue, has side effects that limit dosing, or can’t be administered safely.
BIIB080 took the first failure mode off the table for ARO-MAPT. Lowering tau, in real Alzheimer’s patients, produces measurable effects in the brain and directional cognitive benefits. ARO-MAPT inherits that proof.
What ARO-MAPT still has to prove is that its specific molecule, given as a subcutaneous injection rather than a spinal injection, can match what BIIB080 achieved. That is the question the 2026 readout will answer.
E. The Takeaway
The underlying biology of tau lowering in Alzheimer’s disease has been clinically validated by a competitor. ARO-MAPT inherits that validation. The readout in 2026 is therefore not asking whether tau works as a target. It is asking whether ARO-MAPT, the specific drug, can match what BIIB080 already showed is achievable through a different delivery route.
That is a different question from the one a generic Phase 1/2a candidate faces. It does not guarantee the readout will be positive. ARO-MAPT could still fail, not because tau is the wrong target, but because the specific molecule, the specific delivery, or the specific dose schedule produces a weaker biomarker effect than the trial design requires. Section III addresses why Arrowhead’s subcutaneous transferrin-receptor delivery is structurally well-suited to produce the biomarker effects the readout will measure, and the residual risks are addressed explicitly in the Bottom Line.
For the purpose of this section, the conclusion is narrower and cleaner. The tau target is de-risked. The disease-relevant biology has been demonstrated in patients. The biomarker pathway has been mapped. The cognitive readout has been pattern-matched to approved drugs. ARO-MAPT enters its readout with that foundation already laid.
BIIB080 already proved tau works as a target in Alzheimer’s disease. ARO-MAPT inherits that proof. The question for the 2026 readout is whether ARO-MAPT’s specific molecule, delivered subcutaneously rather than through spinal injection, can match what BIIB080 already showed is achievable. That is a different and easier question than the one most Phase 1 candidates face.
III. Subcutaneous Likely to Beat Intrathecal for Brain Penetration
A. The Bottom Line First
The standard analyst framing of the upcoming ARO-MAPT readout asks whether subcutaneous administration can match what BIIB080 achieved with intrathecal administration. This is the wrong question. It treats spinal injection as the gold-standard delivery route ARO-MAPT must catch up to. The available evidence, from peer-reviewed primate studies, from Arrowhead’s own non-human primate data, and from the basic anatomy of how drugs distribute through the brain, suggests the right question is the opposite. ARO-MAPT is likely to produce deeper, more uniform tau knockdown than BIIB080, particularly in the deep brain regions where Alzheimer’s pathology actually lives.
That is a strong claim. It rests on a structural argument about brain anatomy, on a published comparison of the two delivery approaches in primates, and on Arrowhead’s own primate data showing 70 to 80 percent tau messenger RNA reduction across all 12 brain regions tested, including the brain stem and deep brain structures where intrathecal delivery is weakest. If this argument holds, the implication is significant. It means the 2026 readout could produce a biomarker signal stronger than what BIIB080 produced, not because the underlying tau biology is different, but because the delivery route is fundamentally better suited to the disease’s geography.
This section explains why. It is built around three observations: how the two delivery routes actually distribute drug through the brain, where Alzheimer’s pathology actually lives, and what Arrowhead’s primate data shows about ARO-MAPT specifically.
B. How the Two Delivery Routes Actually Work
The dominant mental model among investors and even some clinicians is that an intrathecal injection (the spinal injection BIIB080 uses) delivers drug “into the brain” the way an IV infusion delivers drug “into the bloodstream.” This is incorrect, and the misconception matters because it obscures the structural asymmetry at the heart of the ARO-MAPT thesis.
When BIIB080 is injected into the spinal column, the drug enters the cerebrospinal fluid, the cushioning fluid that surrounds the brain and spinal cord. From there, it must travel up the spinal column, ascend through the base of the skull, perfuse around the surfaces of the brain, and gradually diffuse inward through the brain tissue to reach the neurons it is supposed to silence. This is a slow, gradient-driven process. Drug concentrations are highest at the brain’s surface and decline steeply as you move toward the deeper structures. The peer-reviewed literature on intrathecal ASO pharmacokinetics in primates, published in part by Ionis researchers themselves, confirms this pattern in detail (see Section III.D for the Mortberg single-cell map and the Frei follow-up characterizing the gradient at deep-brain anatomical resolution).
Subcutaneous delivery works differently. ARO-MAPT is injected under the skin, the way an insulin pen or a GLP-1 weight-loss drug is injected. The drug enters the bloodstream and circulates through the body. To reach the brain, it must cross the blood-brain barrier, the specialized layer of cells that protects the brain from most circulating molecules. Arrowhead’s technology accomplishes this by attaching the drug to a targeting ligand that binds the transferrin receptor protein, a protein that is expressed on the blood-brain barrier and that naturally shuttles molecules from the blood into the brain. The ligand binds the receptor, the receptor pulls the drug across the barrier, and the drug enters the brain tissue uniformly through the cerebrovasculature, the vast network of capillaries that supplies every region of the brain.
The structural difference is fundamental. Intrathecal delivery enters the brain at one place (the surface) and works inward. Subcutaneous transferrin-receptor delivery enters everywhere at once, through the capillaries that supply every tissue region. The two routes have inherently different distribution profiles. Neither is wrong. They are designed for different problems. However, for a target gene that is most consequentially expressed in deep brain structures, which is exactly what Alzheimer’s tau pathology is, the uniform-distribution route has a significant structural advantage.
C. Where Alzheimer’s Pathology Actually Lives
Alzheimer’s disease does not start at the surface of the brain. It starts in the deep medial temporal lobe, a structure called the entorhinal cortex, buried in the inner fold of the temporal lobe, and spreads outward from there in a stereotyped pattern. This pattern was first described by the German neuroanatomist Heiko Braak in the 1990s and is now the canonical staging system for Alzheimer’s pathology in postmortem brain tissue.
The disease progresses through six stages. The first two stages affect only the entorhinal cortex and immediately adjacent structures, some of the deepest, most buried regions of the brain. Stages III and IV expand into the hippocampus and the broader temporal lobe. Stages V and VI eventually reach the cortical surface, where Alzheimer’s pathology is most visible to a clinician but where it is also the latest to arrive. By the time the disease is clinically diagnosed, the deep regions have been accumulating tau pathology for years.
The implication for delivery is direct. The brain regions where Alzheimer’s pathology starts and where it is most pharmacologically consequential are exactly the regions that intrathecal delivery is structurally weakest at reaching. The brain regions where intrathecal delivery is structurally strongest, the cortical surfaces, are the regions where Alzheimer’s pathology arrives last. The two distributions are inverted. A drug that distributes uniformly through the brain via the cerebrovasculature, by contrast, reaches the deep medial temporal regions at the same time and in the same concentration as it reaches the cortical surface.
This is the structural argument at the heart of the ARO-MAPT thesis. BIIB080 has demonstrated that lowering tau works as a therapeutic strategy in Alzheimer’s patients, but BIIB080 is fighting its own delivery route. The drug is being delivered furthest from where the disease lives. ARO-MAPT, if its delivery route works as the primate data suggests, would not have that disadvantage.
D. Independent Corroboration from the Academic Literature
The argument that subcutaneous transferrin-receptor delivery is likely to outperform intrathecal ASO delivery in deep brain tissue is built on two distinct findings in the peer-reviewed literature, each from researchers with no commercial relationship to Arrowhead. The two findings, taken together, independently support both halves of the comparison: that intrathecal delivery is structurally limited in deep brain regions, and that transferrin-receptor-mediated delivery from the bloodstream can effectively reach those regions.
On the limitations of intrathecal delivery, Mortberg and colleagues at the Broad Institute and Ionis Pharmaceuticals (Nucleic Acids Research, 2023) published a single-cell map of ASO activity in mouse and macaque brain following intrathecal administration, characterizing ASO activity across cell types at unprecedented resolution and establishing that knockdown varies by cell type and tissue compartment. A follow-up paper from the same group (Frei and colleagues, 2025) extended this work to three deep brain regions specifically (thalamus, caudate, putamen) and demonstrated the surface-to-deep gradient pattern that constrains intrathecal ASOs in regions farthest from the cerebrospinal fluid. Together these papers represent the most rigorous published characterization of how intrathecal ASO delivery actually distributes in primate brain tissue.
On the feasibility of crossing the blood-brain barrier with transferrin-receptor-conjugated oligonucleotides delivered through the bloodstream, Barker and colleagues (Science Translational Medicine, 2024) published a peer-reviewed study demonstrating that an antibody-conjugated ASO targeting the transferrin receptor produces knockdown in deep brain regions following intravenous administration in non-human primates. The study used a different chemistry from Arrowhead’s ARO-MAPT (the conjugation was antibody-based rather than small-molecule-based) and a different administration route (intravenous rather than subcutaneous), but the underlying mechanism, transferrin-receptor-mediated transcytosis across the blood-brain barrier, is the same. The Barker paper established the modality, that transferrin-receptor-conjugated oligonucleotides can cross the blood-brain barrier from the bloodstream and engage targets in deep brain tissue at clinically relevant levels. Arrowhead’s primate data extends the modality to subcutaneous administration.
Arrowhead’s ARO-MAPT data therefore enters this literature in a particular position. The structural limitation of intrathecal delivery in deep brain regions has been demonstrated independently. The feasibility of subcutaneous transferrin-receptor delivery has been demonstrated independently. Arrowhead is bringing these two findings together with a specific molecule, ARO-MAPT, optimized for the MAPT target. The result is the most uniform deep-brain knockdown pattern published for any oligonucleotide-class drug, characterized in detail across twelve primate brain regions, with cerebrospinal fluid tau reductions sustained for five months. Whether that pattern translates to humans is the question the 2026 readout will answer.
E. What Arrowhead’s Own Primate Data Shows
Arrowhead has now characterized the ARO-MAPT preclinical program in two scientific disclosures at increasing levels of detail. The first, presented by Christine Esau, Ph.D., at the RNA Leaders USA Congress on September 10, 2025, was the first ARO-MAPT-specific dataset and included a direct head-to-head comparison of intrathecal versus subcutaneous siRNA delivery in non-human primates. The second, presented by Agnieszka Glebocka, Ph.D., at the 7th Annual CNS Delivery Summit on December 10, 2025, focused on the subcutaneous platform alone but characterized it in unprecedented detail across twelve brain regions and five months of dosing. Together, the two disclosures establish both the structural advantage of subcutaneous delivery (Figure 2) and the depth and durability of the resulting target engagement (Figures 3 and 4).
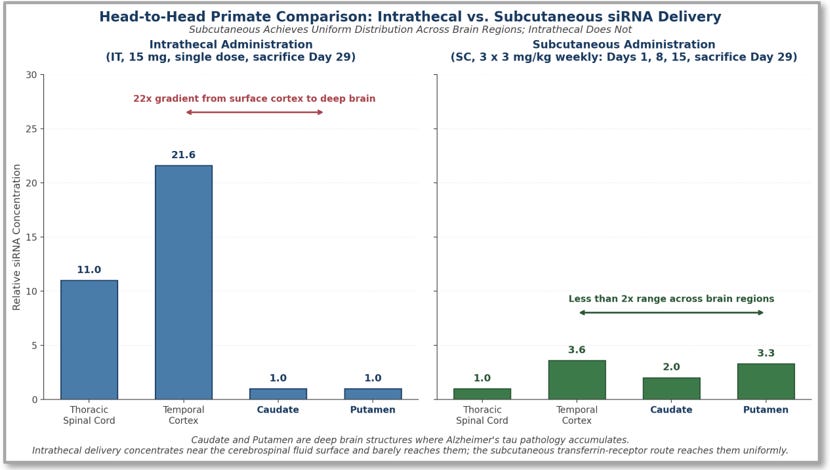
Figure 2. Head-to-head primate comparison of intrathecal (IT) versus subcutaneous (SC) siRNA delivery to four representative brain regions. Intrathecal delivery produces a steep surface-to-deep gradient: high concentrations in the Temporal Cortex (relative concentration 21.6) near the cerebrospinal fluid surface, moderate concentrations in the Thoracic Spinal Cord (11.0), and effectively no delivery to the deep-brain Caudate and Putamen (1.0 each), a 22-fold range. Subcutaneous transferrin-receptor delivery produces uniform distribution across brain regions: 3.6 in Temporal Cortex, 2.0 in Caudate, 3.3 in Putamen (less than a 2-fold range), with the Thoracic Spinal Cord (1.0) the only region where subcutaneous delivers less, consistent with the route’s design. Source: Esau, RNA Leaders USA Congress, September 10, 2025, slide 12.
The most direct way to understand the structural difference between intrathecal and subcutaneous delivery is to look at how each route distributes drug across the same primate brain regions. Figure 2 shows that comparison from the Esau RNA Leaders disclosure. The intrathecal panel shows a steep surface-to-deep gradient: drug concentrations are very high in the Temporal Cortex (21.6) close to the cerebrospinal fluid surface, moderate in the Thoracic Spinal Cord (11.0) which the cerebrospinal fluid bathes directly, and effectively absent in the deep-brain Caudate and Putamen (1.0 each). The subcutaneous panel shows uniform distribution across the brain regions: 3.6 in Temporal Cortex, 2.0 in Caudate, 3.3 in Putamen, less than a 2-fold range, with the Thoracic Spinal Cord (1.0) the only region where subcutaneous delivers less, consistent with the route’s design; subcutaneous transferrin-receptor delivery enters the brain through the bloodstream and bypasses the spinal cord.
A reader might note that intrathecal delivery achieves higher absolute concentrations in the Temporal Cortex (21.6) and Thoracic Spinal Cord (11.0) than subcutaneous achieves anywhere. This is true. The argument the figure makes is not that subcutaneous achieves higher peak concentrations than intrathecal; it is that subcutaneous achieves uniform concentrations across brain regions where intrathecal does not. A delivery platform that hits the surface cortex with twenty-two times the dose of the adjacent deep-brain Caudate and Putamen is not a deep-brain delivery platform; it produces high concentrations near the cerebrospinal fluid surface and effectively none in the structures where Alzheimer’s disease lives. The subsequent slide in the same Esau presentation shows the pharmacological consequence: intrathecal delivery produces minimal MAPT messenger RNA reduction in the deep-brain Caudate and Putamen despite the high cortex concentrations, while subcutaneous delivery produces even MAPT messenger RNA reduction across all four regions. The structural argument from Section III.B has now been documented in primates.
The Glebocka CNS Delivery Summit disclosure, presented three months later, then characterized the subcutaneous platform alone in greater depth. Two figures from that disclosure are particularly consequential.
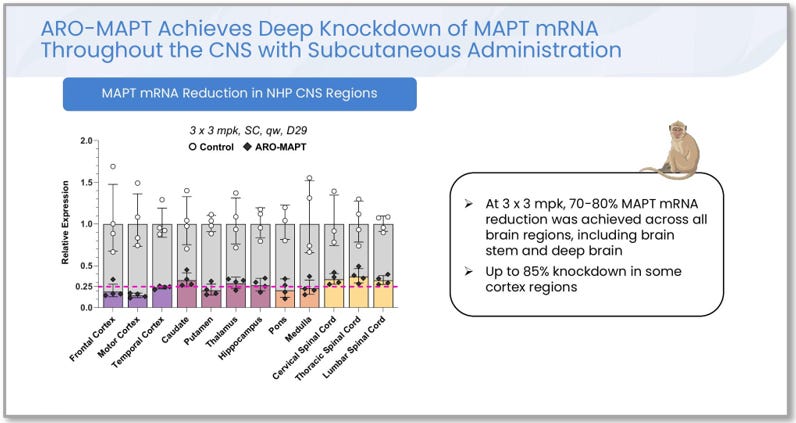
Figure 3. ARO-MAPT MAPT messenger RNA reduction across 12 brain regions in non-human primates, after three weekly subcutaneous doses of 3 mg/kg, measured at Day 29. Reductions of 70 to 80 percent were achieved across all brain regions, including the brain stem and the deep medial temporal structures where Alzheimer’s pathology lives. Up to 85 percent knockdown was achieved in some cortical regions. Source: A. Glebocka, CNS Delivery Summit, December 10, 2025, slide 19.
Three things about this figure matter for the thesis. First, the deep brain regions are not the worst performers. The structures where intrathecal delivery is structurally weakest, the caudate, putamen, thalamus, and hippocampus, all show tau messenger RNA reductions in the 70 to 80 percent range. Second, the brain stem and spinal cord regions, which intrathecal delivery does reach reasonably well, show somewhat lower knockdown than the cortical regions. This is exactly the pattern the structural argument predicts: subcutaneous delivery is engineered for crossing the blood-brain barrier, not for delivering to the spinal cord. Third, the data was measured at Day 29, two weeks after the last of three weekly doses, demonstrating both potency (achieving the knockdown in the first place) and durability (sustaining it for weeks after dosing stopped).
This is consistent with the company’s November 2025 fiscal year-end earnings call, on which CEO Christopher Anzalone described “better than 75% knockdown of tissue-level MAPT mRNA in the CNS” from clinically translatable doses in monkeys. The 70 to 80 percent range disclosed in detail in December 2025 is the more granular scientific version of the same finding.
The third figure is more directly predictive of the human Phase 1/2a readout.
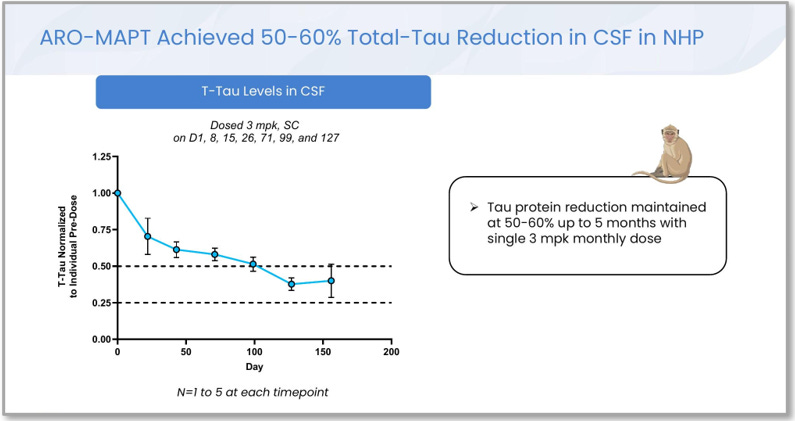
Figure 4. Cerebrospinal fluid total tau reduction in non-human primates dosed with ARO-MAPT at 3 mg/kg subcutaneously: three weekly loading doses (Days 1, 8, 15) followed by approximately monthly maintenance dosing (Days 26, 71, 99, 127). CSF tau reductions of 50 to 60 percent were sustained throughout the five-month dosing window. The CSF tau biomarker is the same biomarker the human Phase 1/2a will measure as its primary pharmacodynamic endpoint. Source: A. Glebocka, CNS Delivery Summit, December 10, 2025, slide 23.
This figure is the most consequential disclosure in the entire ARO-MAPT preclinical package, and it deserves careful attention. The biomarker being measured is cerebrospinal fluid total tau, the same biomarker BIIB080 produced 63 to 67 percent reductions in (Section II), and the same biomarker the human Phase 1/2a in 2026 will measure as its primary pharmacodynamic endpoint. The reductions shown are 50 to 60 percent, sustained for five months in primates, after monthly subcutaneous dosing.
Two implications flow directly. First, the biomarker that the human readout will measure has now been characterized in non-human primates, and the magnitude is in the same range BIIB080 achieved in humans. The 50 to 60 percent range in primates is consistent with a 50 to 70 percent range in humans (primates and humans typically translate at this rate for cerebrospinal fluid biomarkers, sometimes with humans showing somewhat stronger effects due to differences in baseline tau turnover). Second, the durability of the effect, with 50 to 60 percent CSF tau reduction sustained for five months under approximately monthly maintenance dosing in primates, supports the dosing schedule Arrowhead has indicated for the human program: monthly to quarterly subcutaneous injection. This is the same dosing cadence as plozasiran (REDEMPLO), which is approved for quarterly subcutaneous injection.
BIIB080 has shown that lowering tau works as a treatment strategy in Alzheimer’s patients. However, BIIB080 is delivered by spinal injection, which produces drug concentrations that are highest at the brain’s surface and decline as you move toward the deep brain regions where Alzheimer’s pathology actually lives. The drug is fighting its own delivery route.
ARO-MAPT is delivered under the skin, the way an insulin pen is. The drug enters the bloodstream, crosses the blood-brain barrier through a specialized targeting molecule, and reaches every region of the brain through the capillary network. This produces uniform distribution, deep regions and surface regions get the drug in the same concentration.
Arrowhead’s primate data confirms this is happening. Tau messenger RNA reductions of 70 to 80 percent were measured across all 12 brain regions tested, including the deep regions where Alzheimer’s pathology starts. Cerebrospinal fluid tau reductions of 50 to 60 percent were sustained for five months. These are the same kinds of measurements the human Phase 1/2a in 2026 will produce.
F. The Takeaway
The conventional framing of the 2026 readout asks whether ARO-MAPT can match what BIIB080 achieved through spinal injection. The available evidence suggests this is the wrong framing. Subcutaneous transferrin-receptor delivery produces a fundamentally different distribution pattern than spinal injection, and that distribution pattern is better matched to the geography of Alzheimer’s pathology. ARO-MAPT is likely to do more than match BIIB080. It is likely to produce deeper, more uniform tau knockdown in the regions where the disease actually lives.
The structural argument is supported by independent peer-reviewed primate work, on the intrathecal limitation side by Mortberg and colleagues at the Broad Institute (2023), and on the subcutaneous transferrin-receptor feasibility side by Barker and colleagues (2024). It is also supported by Arrowhead’s own primate data, presented in detail at the December 2025 CNS Delivery Summit, showing 70 to 80 percent tau messenger RNA reductions across all brain regions and 50 to 60 percent cerebrospinal fluid tau reductions sustained for five months. The 2026 human readout will test whether this pattern translates from primates to humans.
The right question is not whether ARO-MAPT can catch up to BIIB080. The right question is whether ARO-MAPT can do something BIIB080 structurally cannot, deliver tau lowering uniformly across the deep brain regions where Alzheimer’s pathology actually lives. The available evidence suggests the answer is yes. The 2026 readout will tell us.
IV. Why Phase 1 Is the Catalyst
A. The Bottom Line First
A reasonable reader will accept the case so far and still ask why the 2026 readout matters as an investment catalyst when commercial approval is years away. Why not wait for Phase 2 or Phase 3?
The answer is that the readout will not just validate one drug. It will validate a uniquely valuable platform. Subcutaneous delivery of RNAi therapeutics to the brain has been the most strategically important unsolved problem in the modality for two decades. Arrowhead has now built it. A successful Phase 1/2a readout for ARO-MAPT would be the first human clinical validation of that platform, and the platform itself is what large pharmaceutical acquirers value. The strategic scarcity is the reason Phase 1 is sufficient as a catalyst rather than just a milestone toward a later one.
B. Why Subcutaneous CNS Delivery Is the Holy Grail of RNAi
RNAi produces drugs that work by silencing specific genes. Eight such drugs have been approved by the FDA since 2018, and all eight target genes expressed in the liver, the easiest tissue to deliver to. The brain has been the hardest. The blood-brain barrier exists specifically to keep large molecules out of brain tissue, and RNAi drugs are exactly the kind of large molecule it excludes. For two decades, the entire field has worked on the problem of getting these drugs into the brain at scale. Subcutaneous delivery, the same way an insulin pen is administered, has been the goal but has remained out of reach.
This matters strategically because the brain is where the largest unmet medical needs live. Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, frontotemporal dementia, ALS, the spinocerebellar ataxias, a category of diseases that affect tens of millions of patients globally and for which the drug industry has produced almost nothing that meaningfully changes disease progression. RNAi is in principle ideally suited to most of these diseases, because they are caused by the toxic accumulation of specific proteins, and silencing the gene that produces the protein is exactly what RNAi does. The technology has been ready for years. The delivery has been the wall. A company that solves the delivery problem at scale has access to the largest unmet need in modern medicine.
C. Arrowhead Has Built the Platform, and the Platform Is Now Being Extended
Arrowhead’s central nervous system pipeline has two architectural layers, and the distinction between them is essential to understanding what the 2026 readout actually validates. The first layer is the older intrathecal CNS approach, which delivers drug directly into the cerebrospinal fluid via spinal injection. The most advanced program on this older route is ARO-ATXN2 for spinocerebellar ataxia type 2, partnered with Sarepta Therapeutics under the November 2024 licensing agreement and currently dosing patients in a Phase 1/2 trial in Australia, New Zealand, Canada, Taiwan, and other jurisdictions. The Sarepta partnership covers a broader portfolio that also includes muscle and lung programs, with deal economics of approximately $825 million in initial value plus up to $10 billion in development and commercial milestones across all licensed programs. The CNS programs are not the entire deal, but their inclusion alongside muscle and lung programs is itself a data point: a major pharmaceutical partner saw enough value in Arrowhead’s tissue-targeting capabilities, including but not limited to CNS, to commit at this scale.
The second layer is the newer subcutaneous transferrin-receptor-mediated platform, the platform this paper has been describing throughout. Three programs occupy this layer as of April 2026, with additional programs in earlier development. ARO-MAPT for tau in Alzheimer’s disease and other tauopathies is the first to enter the clinic. The company’s December 2025 press release announcing first dosing explicitly described it as “Arrowhead’s first investigational RNAi-based therapy to utilize a new proprietary delivery system which, in preclinical studies, has achieved blood-brain-barrier penetration and deep knockdown of target genes across the central nervous system, including deep brain regions, after subcutaneous injection.” ARO-SNCA for alpha-synuclein in Parkinson’s disease is the second program on the platform, currently preclinical, with Novartis having taken an exclusive worldwide license in September 2025. SRP-1005, the program formerly designated ARO-HTT and licensed to Sarepta in November 2024, is the third. In January 2026 Sarepta announced submission of a Clinical Trial Application in New Zealand for the INSIGHTT Phase 1 study; Medsafe granted approval in February 2026, and Sarepta has guided to first-in-human dosing in the second quarter of 2026. Sarepta’s public description of SRP-1005 confirms the delivery architecture: “subcutaneous dosing” via “an advanced TfR1 (transferrin receptor protein 1) approach,” “intended to target the deep brain.” Beyond these three, two additional Sarepta-licensed programs are advancing through the platform: ARO-ATXN3 for spinocerebellar ataxia type 3 (preclinical) and ARO-ATXN1 for spinocerebellar ataxia type 1 (discovery), per Arrowhead’s September 2025 disclosure of its CNS portfolio. An additional preclinical program targets an undisclosed cardiometabolic CNS gene referenced as “Gene X” in Arrowhead’s December 2025 CNS Delivery Summit presentation, where ~90 percent target mRNA reduction was demonstrated across all NHP brain regions including deep brain after subcutaneous administration. Counting both architectural layers, the disclosed CNS portfolio comprises seven programs as of early 2026: six on the new subcutaneous transferrin-receptor platform (ARO-MAPT, ARO-SNCA, SRP-1005, ARO-ATXN1, ARO-ATXN3, Gene X) and one on the older intrathecal platform (ARO-ATXN2). Section V develops the implications of the seven-program portfolio in detail.
The strategic implication of this two-layer structure is sharper than the single-layer version of the argument would be. ARO-MAPT is not the most advanced of five interchangeable programs on a proven platform. It is the lead clinical asset on an entirely new platform whose first human validation is its own readout. The 2026 result is not incremental confirmation. It is the first time anyone, anywhere, will have human data showing whether subcutaneous transferrin-receptor delivery of an RNAi therapeutic produces target engagement in the central nervous system. SRP-1005 will follow with its own Phase 1 readout on a related but independent target, providing a second platform read at a similar moment in time. The two readouts together will, in combination, either validate the platform across multiple targets or call its translation into question. ARO-MAPT goes first.
Two facts make this binary less binary than it may first appear. First, the underlying mechanism, transferrin-receptor-mediated transcytosis across the blood-brain barrier, has been independently validated in non-human primates by Denali Therapeutics (Barker et al., 2024, Section III.D), so the modality itself is not in question. The 2026 readout is asking whether Arrowhead’s specific molecule on its specific delivery chemistry produces the cerebrospinal fluid tau reductions the primate data predicts. Second, multiple Big Pharma diligence processes have already concluded the platform is worth substantial money at the preclinical stage. Novartis’s September 2025 license of ARO-SNCA for $200 million upfront and up to $2 billion in milestones, and Sarepta’s broader $825 million-plus collaboration covering ARO-HTT (now SRP-1005) and related programs, are independent diligence-stage validations of what the platform could be worth once human data exists. Diligence judgments are not clinical data, but they are also not nothing.
D. The Clinical Lead Is the Moat, and Big Pharma Knows It
Other companies in the RNAi and oligonucleotide space have approached central nervous system delivery, but none has reached comparable clinical advancement. Alnylam Pharmaceuticals, the largest RNAi company by revenue and approved drugs, has all six of its approved drugs targeting genes expressed in the liver; its central nervous system efforts use C16 lipid conjugates that are administered intrathecally, not subcutaneously. Atalanta Therapeutics is developing branched divalent siRNA scaffolds (di-siRNA) for Huntington’s disease and other central nervous system targets, but its delivery route is also intrathecal/cerebrospinal fluid administration rather than subcutaneous. Ionis Pharmaceuticals is the closest competitor in central nervous system oligonucleotide delivery overall, but Ionis uses ASO chemistry rather than RNAi, and its approved central nervous system drugs (nusinersen for spinal muscular atrophy and tofersen for SOD1 ALS) all use intrathecal delivery. Denali Therapeutics has demonstrated transferrin-receptor-mediated ASO BBB crossing in primates (Barker et al., 2024) but has not advanced an oligonucleotide-conjugate CNS program into the clinic; Denali’s clinical CNS work to date has used antibody-conjugated enzymes (DNL310 for MPS II, DNL126 for MPS IIIA, DNL952 for Pompe) rather than RNA-class therapeutics. Wave Life Sciences uses a different oligonucleotide chemistry, and Wave’s subcutaneous siRNA work has been demonstrated for hepatic targets, not for central nervous system targets. Switch Therapeutics, City Therapeutics, and other newer entrants in the broader RNAi space remain at preclinical stages and have not announced subcutaneous CNS clinical programs.
The most relevant competitive disclosure is the most recent. On April 27, 2026, Dyne Therapeutics announced preclinical data from its FORCE platform showing approximately 75% MAPT mRNA knockdown in non-human primates using a transferrin-receptor-binding antibody fragment conjugated to MAPT siRNA, with widespread and consistent delivery across brain regions including the deep brain. Dyne also confirmed equivalent MAPT mRNA reduction with subcutaneous and intravenous administration in mice. This is the first competitive disclosure from a non-Arrowhead-licensee that addresses the same mechanistic configuration (transferrin-receptor-mediated subcutaneous siRNA-to-CNS) that Arrowhead’s platform addresses. Several aspects of the Dyne disclosure warrant precision. The 75% knockdown is reported for an optimized FORCE Fab variant (Conjugate 2); the original clinically validated FORCE Fab (Conjugate 1) was shown active in NHP but its knockdown magnitude was not quantified in the announcement. The press release confirmed subcutaneous-equivalent activity in mice but did not specify whether the NHP study itself used subcutaneous or intravenous administration. Dyne disclosed mRNA knockdown only; no protein-level effects, durability data, or tolerability data were reported. Most importantly, the program is preclinical and Dyne explicitly stated it is “evaluating potential next steps for the preclinical development of these conjugates with the goal of maintaining capital efficiency and maximizing shareholder value,” which in industry context indicates the program is being positioned for partnership rather than internal clinical advancement. Dyne’s primary commercial focus remains Duchenne muscular dystrophy and myotonic dystrophy type 1, where its lead clinical assets are advancing.
The Dyne disclosure has two implications, and they cut in opposite directions. The first is that the underlying transferrin-receptor-mediated subcutaneous siRNA-to-CNS modality has now been independently demonstrated in non-human primates by a second platform, which strengthens confidence in the modality at the mechanism level. The biology works. The route of administration is feasible. Tau as a target is reachable. These are confirmations of what Arrowhead’s preclinical package had already suggested. The second implication is that the field is no longer literally empty outside the Arrowhead-licensee ecosystem. The accurate framing is that as of April 2026, Arrowhead has the only clinical-stage subcutaneous RNAi CNS program in development, with ARO-MAPT dosing patients since December 2025 and the Phase 1/2a readout expected in the second half of 2026. Dyne’s preclinical-stage program would, even on an aggressive development timeline, require approximately two to three years to reach human dosing if Dyne or a future partner chose to advance it. The clinical lead is years, and clinical leads in regulated drug development compound rather than decay. Each additional month of human data Arrowhead generates while competing platforms remain in preclinical evaluation is an additional month of widening moat.
This scarcity creates two layers of acquisition pressure on potential pharmaceutical bidders. The first is the obvious one: any pharmaceutical company that wants near-term subcutaneous central nervous system RNAi capability has, today, a single credible target to acquire to get it. Any longer-term competitor, including a future-licensed Dyne program, would arrive at clinical validation years later, behind a platform with established intellectual property, accumulated human data, and entrenched commercial relationships. That alone supports premium valuations.
The second layer is more important and is rarely discussed explicitly in research notes. Pharmaceutical companies are intensely strategic about platform technologies, and they do not just want platforms for themselves, they specifically do not want their competitors to control them. A pharmaceutical company that lets a rival acquire Arrowhead would not just lose the chance to acquire the platform; it would watch a competitor secure a multi-decade durable advantage in central nervous system drug development, in the disease category with the largest unmet need in modern medicine. The defensive logic compounds the offensive logic. Once Arrowhead’s platform is clinically validated by the 2026 readout, the question for each potential acquirer becomes not just whether to bid, but how much it would cost to let someone else win. Competitive auctions driven by both offensive and defensive logic produce the highest acquisition prices in pharmaceutical mergers and acquisitions, and the dynamic is exactly what The Endgame paper develops.
E. What This Means for the 2026 Readout
The 2026 Phase 1/2a readout is the catalyst because it converts Arrowhead’s subcutaneous central nervous system platform from preclinically promising to clinically validated. Two external data points already speak to what that conversion is worth. First, in September 2025 Novartis announced a global licensing agreement for ARO-SNCA, the preclinical Parkinson’s program on the SC transferrin-receptor platform. The deal terms: $200 million upfront, plus up to $2 billion in development, regulatory, and sales milestones, plus tiered royalties on commercial sales. The deal closed in October 2025. Critically, Novartis committed at this scale before any human data existed for the platform. Second, Sarepta’s January–2026 CTA submission and February–2026 CTA approval for SRP-1005 (formerly ARO-HTT) means a second major pharmaceutical sponsor has now committed to running its own Phase 1 study on the same subcutaneous transferrin-receptor delivery architecture, with first-in-human dosing planned for the second quarter of 2026. The implied valuation per program from each of these transactions reflects diligence-stage assessment of platform risk, not validated platform risk. The price of clinical validation, when it arrives, will sit on top of the price of preclinical validation, not next to it.
The investor implication is that the readout is sufficient to trigger material reassessment of Arrowhead’s value, through whichever mechanism eventually delivers it, stock price re-rating, partnership announcements, milestone payments, or outright acquisition. The reader is not being asked to predict which mechanism. The reader is being asked to weigh whether a position at current prices, before the catalyst, is justified by the structure of the evidence.
F. The Takeaway
Subcutaneous delivery of RNAi drugs to the brain has been the largest unsolved problem in the modality for two decades. Arrowhead is the company that built the platform that solves it. ARO-MAPT is the first drug on that platform to enter the clinic, with first-in-human dosing in December 2025 and the Phase 1/2a readout expected in the second half of 2026. SRP-1005, licensed to Sarepta, is the second drug on the platform to enter the clinic, with dosing planned for the second quarter of 2026. ARO-SNCA, licensed to Novartis, is the third program on the platform, preclinical, with a $200 million upfront and up to $2 billion in milestones already paid against the diligence-stage assessment of platform risk. As of April 2026, Arrowhead has the only clinical-stage subcutaneous RNAi CNS program in development; the most recent independent preclinical disclosure (Dyne Therapeutics, April 27, 2026, Section IV.D) is several years from clinical entry and is being positioned by Dyne for partnership rather than internal advancement. The 2026 ARO-MAPT readout is the first human clinical validation event for the platform. The platform itself is what large pharmaceutical acquirers value, both for what it gives the acquirer and for what it denies the competition.
The 2026 ARO-MAPT readout will not just validate one drug. It will be the first human test of an entirely new modality, the first subcutaneous transferrin-receptor delivery of an RNAi therapeutic to be tested in patients, in the disease area with the largest unmet need in modern medicine. SRP-1005, on the same platform, will follow with its own Phase 1 readout shortly after. Once human data exists, every potential pharmaceutical acquirer faces both the offensive question (how much is the platform worth to us) and the defensive question (how much is it worth to keep it from a competitor). Novartis’s September 2025 decision to pay $200 million upfront plus up to $2 billion in milestones for a single preclinical asset on the platform, and Sarepta’s broader collaboration covering SRP-1005 and related programs, already tell us what the diligence-stage assessment looks like. The price of clinical validation, when it arrives, will sit on top of that. The Endgame develops the resulting valuation scenarios in detail.
V. Signs that Arrowhead Knows What It Has
A. The Bottom Line First
Companies tell you what they believe by what they retain, where they stake their next bets, and where they pre-position their physical capacity. By every observable signal, Arrowhead is acting like a company that has already concluded what the 2026 readout will show. Leadership has positioned the company to execute when the readout arrives.
The argument in this section is not that the readout is more likely to succeed because management is acting confidently. Confidence does not produce data. The argument is narrower and cleaner. An investor who concludes the platform is uniquely valuable is reaching the same conclusion management has already reached, and is doing so before the public market repricing that the 2026 readout will trigger.
B. The Verona Facility and the 6.24-Acre Land Option
Arrowhead’s Verona, Wisconsin facility is a validated, FDA-inspected GMP manufacturing operation with three parallel production lines capable of supporting nine clinical programs simultaneously. As The Endgame establishes in detail in its Section XIII, Arrowhead also holds an option to acquire 6.24 acres of land directly adjacent to the Verona campus, with the option expiring in 2028.
A clarification on what this option does and does not signal is in order. The option itself predates ARO-MAPT and predates the broader subcutaneous CNS delivery platform. It was secured as general manufacturing optionality supporting Arrowhead’s broader platform strategy rather than as a response to any specific clinical asset. Its existence is therefore not by itself evidence of MAPT-specific confidence. What matters is what Arrowhead does with the option in 2026 and 2027.
The timing alignment is the key fact going forward. The 2H 2026 ARO-MAPT readout sits roughly eighteen months ahead of the option expiration. If the readout produces the cerebrospinal fluid tau reductions the primate data predicts, Arrowhead will need to make a binding decision on commercial-scale manufacturing capacity well before the option lapses. The expected behavior, if the readout succeeds, is exercise. If the readout disappoints, one may expect the option to lapse with the existing facility to continue supporting the pipeline at current scale.
This makes the option a forward-looking signal rather than a backward-looking one. The reader should watch for the exercise decision in 2026 or 2027 as a specific revealed-preference signal of management’s response to the data. Exercise would constitute the most explicit form of capital commitment Arrowhead can make in response to the readout: bigger than a hire, more durable than a partnership term, less reversible than a capital raise. The full acquisition-currency analysis, including the $1 to $3 billion in combined facility-and-option value to a strategic acquirer, is developed in The Endgame.
C. The Partnership Retention Pattern
Section IV develops the offensive valuation signal embedded in Arrowhead’s two large partnership transactions: $200 million upfront and up to $2 billion in milestones from Novartis for ARO-SNCA in September 2025; over $825 million in initial value plus up to $10 billion in milestones from Sarepta for SRP-1005 (formerly ARO-HTT) and related programs in November 2024. Those numbers tell the reader what Big Pharma is willing to pay for diligence-stage access to programs on the platform.
The same transactions tell a different and complementary story when read in reverse. What Arrowhead retained matters as much as what Arrowhead sold. ARO-MAPT, the lead asset in the largest indication (Alzheimer’s disease), is wholly owned by Arrowhead. The broadest tauopathy indications are wholly owned by Arrowhead. The therapeutic areas the platform may unlock as new tissue-targeting capabilities mature, including additional CNS targets beyond the partnered set, the obesity programs, the ocular and cardiomyocyte programs, are wholly owned by Arrowhead.
The retention pattern is the behavior of a company that has internalized the platform’s strategic value and is selling at the periphery while holding the core. A company that believed the lead asset was worth what the partnered programs imply would not retain it. It would license it on similar terms and capture the upfront economics, the way a typical pipeline company monetizes its best-in-class asset. Arrowhead did not.
D. Pre-Validation Target Staking
The strategic logic for a platform company that believes its platform works is to stake as many high-value targets as possible during the period before public validation. The reason is structural. Arrowhead’s manufacturing lead is durable: new commercial-scale oligonucleotide facilities take five to seven years to build, which is roughly the lead time the company has banked. Target staking is a separate and faster race. The moment human data confirms the subcutaneous CNS delivery platform works, every Big Pharma neurology team and every well-funded RNAi specialist will redirect resources toward CNS targets that Arrowhead has not already claimed. The cheap window for staking is now.
Arrowhead has been executing this strategy. The publicly disclosed CNS portfolio includes seven programs as of early 2026: ARO-MAPT (Alzheimer’s and tauopathies, wholly owned, lead clinical asset); ARO-HTT, now SRP-1005 (Huntington’s disease, partnered with Sarepta, dosing planned for 2Q 2026); ARO-SNCA (Parkinson’s and synucleinopathies, partnered with Novartis, preclinical); ARO-ATXN1 (spinocerebellar ataxia type 1, partnered with Sarepta, discovery); ARO-ATXN2 (spinocerebellar ataxia type 2, partnered with Sarepta, Phase 1 intrathecal); ARO-ATXN3 (spinocerebellar ataxia type 3, partnered with Sarepta, preclinical); and an undisclosed cardiometabolic CNS target referenced as “Gene X” in Arrowhead’s December 2025 CNS Delivery Summit presentation, where ~90 percent target mRNA reduction was demonstrated across all NHP brain regions including deep brain after subcutaneous administration. The fact that Arrowhead disclosed primate efficacy data on Gene X without disclosing the target itself indicates that the program is being held proprietary, likely because the target is novel and competitively sensitive. Five of the seven disclosed programs are partnered. The structure of those partnerships is itself revealing.
The Sarepta deal in November 2024 explicitly covered four CNS targets in a single transaction (HTT, ATXN1, ATXN2, ATXN3), plus muscle and lung programs. The Novartis deal in September 2025 included not only ARO-SNCA but also options on a limited number of additional collaboration targets outside Arrowhead’s pipeline, to be developed using the TRiM platform. Arrowhead is not just selling individual assets. The company is selling target-staking access to its platform, on terms structured to protect the lead asset and keep the discovery engine in-house. This is what a platform company does when it has more high-value targets in front of it than it can advance alone, and wants to lock them up before competitors can.
The broader CNS opportunity space is real and being pursued. Other companies have demonstrated that CNS protein-lowering through oligonucleotide therapeutics is therapeutically valuable: tofersen for SOD1-ALS, the first such drug approved; BIIB080 for tau (Section II); Wave Life Sciences’ prior C9orf72 program; Ionis’s ongoing PRNP program for prion disease; and multiple academic and industry programs targeting GBA1, KCNT1, SCN1A, SCN8A, APP, TDP-43, and additional aggregation-prone CNS proteins. Each is currently being pursued through intrathecal delivery, the structurally inferior route established in Section III. Once Arrowhead’s subcutaneous platform is clinically validated, every one of these targets becomes addressable through a delivery route that is patient-acceptable rather than burdensome, with broader brain distribution than intrathecal can achieve. Arrowhead is structurally advantaged to compete for any of them. The cheap staking window is the period before that advantage becomes broadly priced into the competitive landscape.
The forward question is whether the pace continues. If management’s behavior reflects their internal probability assessment of the readout, expect additional CNS targets to be disclosed before the data arrives. Likely vehicles: new preclinical programs unveiled at industry conferences, expanded partnership terms that pull additional targets under existing deals, or a new partnership entirely. Additional CNS programs disclosed in 2026 ahead of the readout would be another concrete revealed-preference signal of the same kind. The window for cheap staking closes the day the readout is announced.
Companies tell you what they believe by what they do, not by what they say. By that standard, Arrowhead has been telling the story of an enormously valuable platform since well before the 2026 readout could confirm it.
The partnership deals that look generous to Novartis and Sarepta are conspicuously absent the lead asset, ARO-MAPT, which Arrowhead kept. Seven CNS programs have already been staked (six named plus an undisclosed cardiometabolic CNS target), with partnership structures that pull additional targets under platform terms. And a 6.24-acre land option adjacent to Verona expires in 2028, meaning the most concrete capital decision of all sits in management’s hands roughly eighteen months after the readout.
Investors who conclude the platform is uniquely valuable are arriving at the same conclusion management has already reached. The behavior is the evidence.
E. The Takeaway
The behavioral evidence assembled in this section does not change the probability of a successful 2026 readout. What it does is establish that Arrowhead’s leadership has been positioning the company for the readout’s success across the dimensions where its conviction is most observable: a partnership structure that retains the lead asset while monetizing the periphery, a CNS portfolio that has staked seven programs before any human validation exists, and a Verona land option whose exercise decision sits in management’s hands roughly eighteen months after the readout. Each is consistent with the same internal conclusion. The platform is uniquely valuable, and the 2026 readout will validate that.
The Verona land option occupies a particular epistemic role. The option itself predates the subcutaneous CNS platform and does not speak directly to MAPT-specific confidence. The exercise decision in 2026 or 2027, however, will. Watch for it. Exercise would be the most concrete demonstration of management’s response to the data that Arrowhead can produce, and it would arrive in a form that no analyst report and no earnings call language can match: dirt, equipment, and a permanent expansion of the company’s commercial-scale manufacturing footprint.
The investor’s job between now and the readout remains the same as it was at the end of Section IV: weigh the evidence and decide whether the structure of the case justifies a position before the readout resolves the question. This section adds one more piece to that weighing. The company is acting as if it has already drawn its conclusion.
VI. The Bottom Line
ARO-MAPT is not a generic Phase 1 candidate. The tau target has already been clinically validated by BIIB080, which produced 63 to 67 percent cerebrospinal fluid tau reductions in 46 mild Alzheimer’s patients and directional cognitive trends matching the approved amyloid antibodies. The delivery route, subcutaneous injection through a transferrin-receptor-mediated platform, distributes drug uniformly across the brain rather than producing the surface-to-deep gradient that intrathecal injection produces. This matters because Alzheimer’s pathology starts in the deep medial temporal lobe, where intrathecal delivery is structurally weakest. Arrowhead’s primate data shows 70 to 80 percent MAPT messenger RNA reductions across all twelve brain regions tested, with deep brain regions performing as well as cortical regions, and 50 to 60 percent cerebrospinal fluid tau reductions sustained for five months.
The Phase 1/2a trial measures cerebrospinal fluid tau as its primary pharmacodynamic biomarker, the same biomarker BIIB080 lowered, the same biomarker Arrowhead’s primate program characterized, and the same biomarker the human readout will produce. The 2026 readout will either show ARO-MAPT produced the cerebrospinal fluid tau reductions the primate data predicts, or it will not.
The argument is not that ARO-MAPT cannot fail. It can. The primate-to-human translation could disappoint. The Phase 1/2a is a small study, and inter-subject variability in cerebrospinal fluid biomarkers is substantial. The track record of anti-tau drugs in Alzheimer’s disease is poor, although the failed programs targeted extracellular tau or worked through unrelated mechanisms, and ARO-MAPT operates upstream of those failures. Each of these risks is real. None of them changes the underlying structure of the evidence. The argument is that the evidence assembled across the prior sections justifies a meaningful conviction in the readout, well above the generic Phase 1 base rate, while leaving the residual risk to be sized by each reader’s own conviction.
The 2026 readout will deliver the verdict. The investor’s job between now and then is to weigh whether the preclinical and clinical evidence already assembled: four progressively detailed scientific disclosures over two and a half years, the BIIB080 clinical validation of the underlying biology, the academic literature characterizing intrathecal’s gradient limitation and transferrin-receptor delivery’s feasibility for crossing the blood-brain barrier, the structural anatomy argument about where Alzheimer’s lives in the brain, and the partnership pricing that establishes diligence-stage Big Pharma valuation, corroborated by the revealed-preference behavior of Arrowhead’s leadership, justifies a position before the readout resolves the question. This paper has assembled that evidence as completely as the available data allows. The conclusion is the reader’s to draw.
A Note on Supporting Independent Research
These white papers took hundreds of hours to produce. The asset inventory, valuation methodology, bidder analysis, comparable transaction work, acquisition thesis, competitor analysis, supporting charts, and science analytics are the result of deep primary research and is not available in sell-side coverage. Most of the analysis presented represents independent research not published elsewhere. It is being shared freely because the thesis deserves the widest possible audience. If ARO-MAPT delivers in 2H 2026, every Arrowhead shareholder benefits from a well-informed market that understands what the data means and what the asset is worth. That is why this paper exists.
If this research has been valuable to you, whether it shaped your thinking, validated your conviction, or simply saved you the time of doing this work yourself, a voluntary contribution is genuinely appreciated and directly funds the next paper.
For individual investors and readers
Any amount you feel reflects the value you received is welcome and meaningful. A contribution in the range of what you might pay for a single premium research report is a thoughtful gesture that makes a real difference.
For family offices, investment funds, hedge funds, and research platforms
This paper is the caliber of work that institutional research desks bill significant retainers to produce. If your team referenced it, distributed it internally, or used it to inform a position, a suggested contribution of $1,500 or more reflects the professional value of the analysis, though any amount is meaningful. Your support makes it possible to continue publishing at this level without a paywall that limits the reach of the ideas. If your organization requires an invoice to process a payment, please reach out directly at bioboyscout@gmail.com and one will be provided promptly.
There is no obligation and no expectation. This is purely a thank you for work that meant something to you.
Zelle: (847) 227-7909
Thank you for reading, and for being part of a community that takes this thesis seriously.
— Robert Toczycki | BioBoyScout
Important Risks, Disclosures, & Disclaimers
The author, Robert Toczycki (aka BioBoyScout), certifies that:
all views expressed in this white paper accurately reflect his personal opinions about the topic discussed; and
he was not compensated in any form for producing this white paper.
This white paper is published by BioBoyScout and is intended for informational and educational purposes only. It does not constitute investment advice, a solicitation to buy or sell securities, or a guarantee of future results. The author holds a long position in Arrowhead common stock. Arrowhead Pharmaceuticals (ARWR) is a publicly traded company; investments in its shares involve material risks, including the risk of total loss. All financial projections, acquisition price estimates, and valuation analyses herein are hypothetical frameworks for analytical purposes and do not represent predictions of actual outcomes. Readers should conduct their own due diligence and consult a registered investment advisor before making investment decisions. All data cited herein were sourced from publicly available company disclosures, SEC filings, press releases, and peer-reviewed literature as of May 2026.
About the Author
Robert Toczycki is an independent analyst and registered US Patent Attorney with a JD, an Executive MBA completed at the top of his class, and a BS in Mathematics and Computer Science from the University of Illinois at Urbana-Champaign. He has a deep passion for financial analysis, particularly identifying valuation discrepancies and demonstrating them through rigorous, data-driven research and solid analytics.
Comments or questions: bioboyscout@gmail.com.
Copyright © 2026, BioBoyScout. All Rights Reserved.