The Year of Tau
ARO-MAPT, the CNS Delivery Breakthrough, and Why 2H 2026 Could Redefine Arrowhead Pharmaceuticals
Originally published April 8, 2026, as a BioBoyScout white paper. Republished here on Substack with full content, embedded charts, and downloadable PDF. — Robert
A Deep-Dive Investment Thesis on Subcutaneous CNS Delivery, ARO-MAPT vs. the Intrathecal Field, the Tauopathy Indication Ladder from PSP to Alzheimer's, the Ocular and Cardiomyocyte Platform Frontiers, and Why the Street Is Systematically Mispricing the Most Important Platform Inflection in Arrowhead's History
Robert Toczycki, JD, MBA
bioboyscout@gmail.com
847.227.7909
X: @BioBoyScout
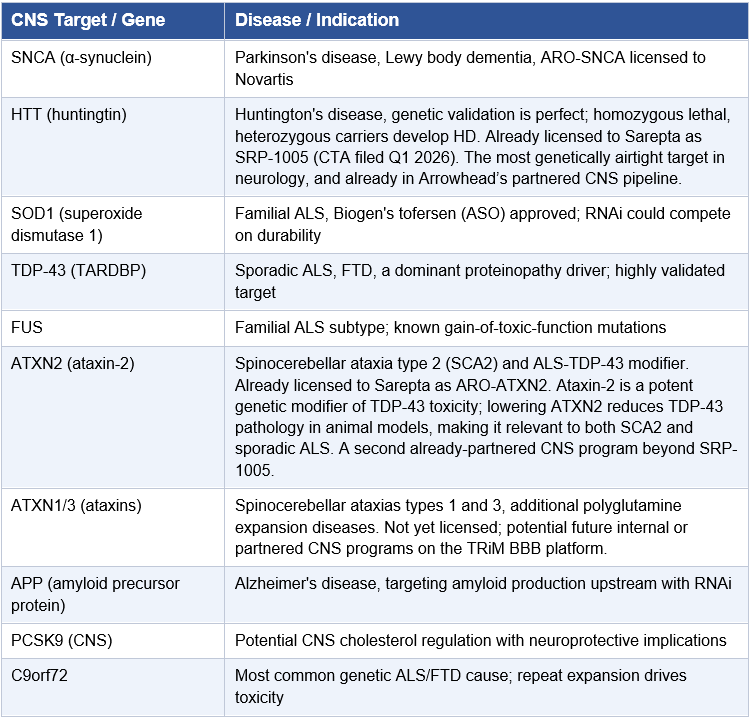
Arrowhead Pharmaceuticals (NASDAQ: ARWR) has spent a decade systematically validating its TRiM™ platform across the liver, solid tumors, lung, muscle, adipose tissue, and now, the brain. While the CNS program is the headline, Arrowhead has also signaled its next tissue frontiers: the eye and the heart. Preclinical data on a trabecular meshwork-targeted TRiM platform for glaucoma, delivered via intracameral injection, have already been presented at scientific conferences. The pattern is unmistakable: CNS is the immediate inflection, and ocular and cardiomyocyte are next in line. This paper focuses on CNS, specifically ARO-MAPT and what 2H 2026 data means for the stock, but the ocular and heart expansions reinforce the same underlying thesis: Arrowhead is not a pipeline company. It is a delivery platform company systematically conquering every major tissue in medicine, and no competitor comes close to matching its breadth across extrahepatic targets.
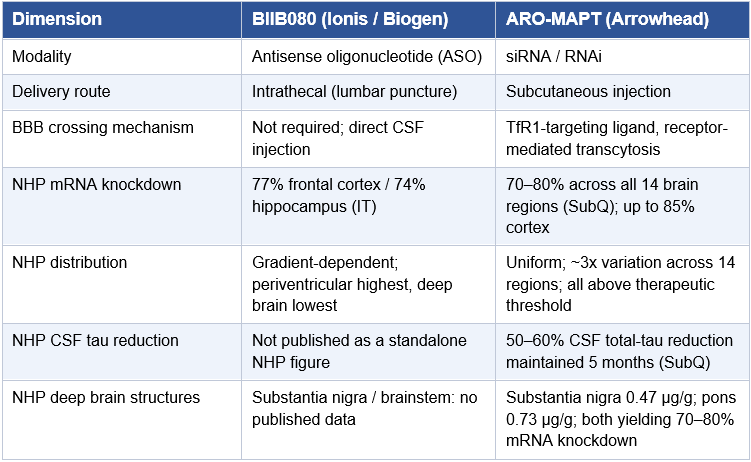
Key Findings at a Glance
The following table summarizes the most important data points, benchmarks, and analytical conclusions in this paper. Readers are encouraged to use this as a navigational reference before proceeding to the full analysis.
ARO-MAPT NHP mRNA knockdown
70–80% MAPT mRNA reduction across all 14 measured brain regions; up to 85% in some cortex regions. Achieved via subcutaneous injection at clinically translatable doses. Source: 7th Annual CNS Delivery Summit, December 10, 2025.
ARO-MAPT NHP CSF tau reduction
50–60% total-tau protein reduction in NHP CSF, maintained for up to 5 months with monthly subcutaneous dosing. This is the most critical preclinical figure: it matches BIIB080’s best human Phase 1b result via the same biomarker endpoint.
BIIB080 NHP benchmark
74–77% MAPT mRNA knockdown in frontal cortex and hippocampus via intrathecal injection. Translated to 56–60% CSF total-tau reduction in humans (Mummery et al., 2023). This is the reference standard.
ARO-MAPT human data projection
Central estimate: 40–55% CSF total-tau reduction at optimal loading dose in 2H 2026 initial readout. Derived from ARO-MAPT NHP CSF tau result and BIIB080 NHP-to-human translation ratio (~73–78% retention).
Deep brain distribution advantage
ARO-MAPT siRNA trigger concentration ranged 0.47–1.55 μg/g across 14 brain regions (~3x variation). Substantia nigra, pons, and medulla all produced 70–80% knockdown. BIIB080 has no published NHP knockdown data for these structures.
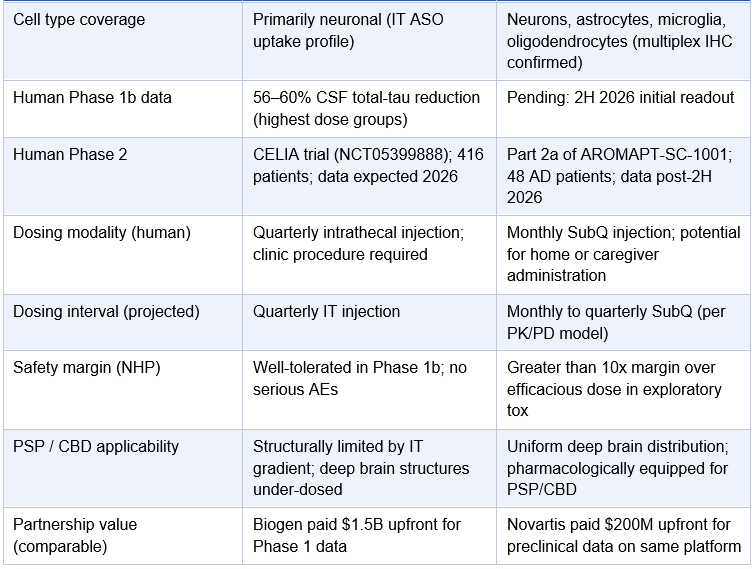
Cell type coverage
ARO-MAPT achieves MAPT mRNA knockdown across neurons, astrocytes, microglia, and oligodendrocytes in NHP brainstem (RNAscope multiplex IHC confirmed). Relevant to CBD and PSP where glial tau pathology is prominent.
Novartis precedent valuation
$200 million upfront for ARO-SNCA, a single preclinical CNS program on the same delivery platform. Established before any human data. Post-validation partnership terms would be materially higher.
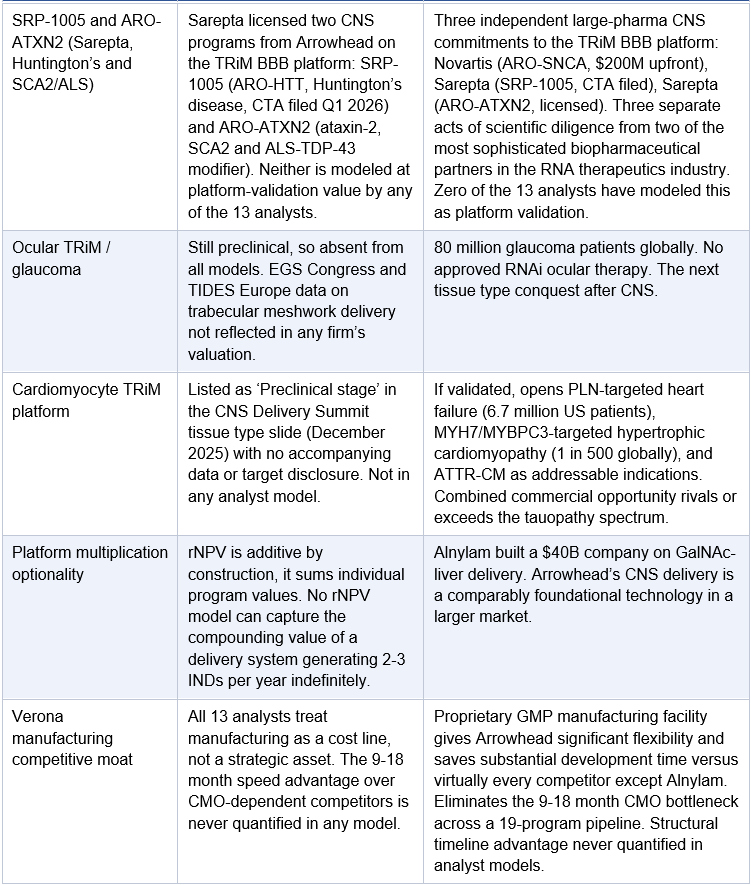
Sarepta CNS programs: SRP-1005 and ARO-ATXN2
Sarepta filed a CTA for SRP-1005 (ARO-HTT, Huntington’s disease) in Q1 2026 using the same TRiM BBB delivery system. Arrowhead has also licensed ARO-ATXN2 (ataxin-2, targeting SCA2 and ALS) to Sarepta. Two Sarepta CNS programs plus Novartis ARO-SNCA means three separate large-pharma programs already on the TRiM BBB platform, none of which appear in sell-side models at platform-level value.
Safety margin
Greater than 10x safety margin over efficacious dose confirmed in rodent and NHP exploratory toxicology. No dose-limiting toxicities in either species.
Current market pricing of ARO-MAPT
Approximately zero contribution to ARWR’s current ~$8 billion market capitalization. ARO-MAPT is not in any of the 13 official sell-side models at platform-level value.
BIIB080 CELIA Phase 2
Phase 2 trial fully enrolled April 2025 (416 patients). Efficacy data expected 2026. Could report before or after ARO-MAPT Phase 1 data. Both outcomes are analytically constructive for ARO-MAPT.
Key risk
NHP-to-human BBB translation is not guaranteed. TfR1 expression is confirmed cross-reactive between cynomolgus macaques and humans, but the human BBB has structural differences. Phase 1 human data is the decisive test.
I. Executive Summary
The central claim of this paper is simple: ARO-MAPT is not just a drug candidate. It is the clinical proof-of-concept test for Arrowhead’s subcutaneous CNS delivery technology, a technological achievement that, if validated in humans, would be one of the most significant developments in the history of RNAi therapeutics.
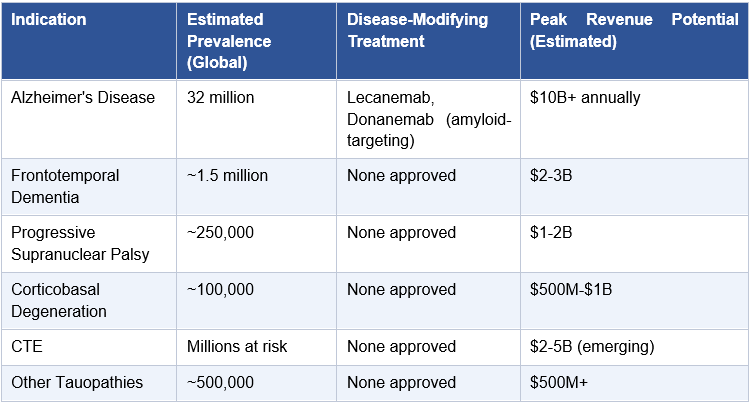
Alzheimer’s disease and the broader tauopathy spectrum represent the largest unmet medical need in medicine. Over 32 million people worldwide live with Alzheimer’s. The amyloid hypothesis has dominated the field for three decades, and while lecanemab (Leqembi) and donanemab (Kisunla) have provided proof that amyloid clearance can modestly slow disease progression, the field increasingly recognizes that tau, not amyloid, is the protein most tightly correlated with cognitive decline and neurodegeneration. This relationship is reviewed in detail in Lleo et al. (Cell, 2025), which describes the evolving Alzheimer’s therapeutic landscape and the growing case for tau as the executor of disease progression. Tau is the next frontier.
Arrowhead dosed its first subjects in the AROMAPT-SC-1001 Phase 1/2a trial on December 8, 2025. The trial will evaluate ARO-MAPT in up to 64 healthy volunteers and 48 patients with early Alzheimer’s disease. Initial data are expected in the second half of 2026. Critically, ARO-MAPT uses a novel subcutaneous delivery mechanism that, in non-human primate studies at clinically translatable doses, achieved 70–80% MAPT mRNA knockdown across all brain regions including deep brain structures, with up to 85% knockdown in cortex. More importantly, it then translated that mRNA reduction into 50–60% CSF total-tau protein reduction maintained for up to 5 months with monthly subcutaneous dosing. These figures were first presented publicly at the 7th Annual CNS Delivery Summit on December 10, 2025. That CSF protein result is already in the same range as BIIB080’s best human Phase 1b outcome (56–60% CSF tau reduction), achieved by Biogen via lumbar puncture in human Alzheimer’s patients. ARO-MAPT has already achieved in NHPs, subcutaneously, what took BIIB080 a human Phase 1 trial to demonstrate. This is the first Arrowhead program to use this CNS delivery system.
The competitive landscape is anchored by BIIB080 (ION464), the Ionis/Biogen antisense oligonucleotide (ASO) that achieved approximately 60% tau reduction in CSF biomarkers in Phase 1/2 and whose Phase 2 CELIA trial (NCT05399888) was fully enrolled in April 2025 with efficacy data expected in 2026. BIIB080’s existence validates the tau reduction hypothesis and establishes the biomarker benchmarks ARO-MAPT must meet. But ARO-MAPT has the potential to be a generational improvement over BIIB080 in one critical dimension: delivery modality. Subcutaneous injection at home vs. repeated intrathecal lumbar punctures in a clinic is not a marginal quality-of-life difference. It is the difference between a scalable therapy and a niche one.
The investment case for ARWR on the back of positive ARO-MAPT data rests on three interlocking theses:
(1) the drug works: human tau knockdown is demonstrated, validating the therapeutic mechanism;
(2) the delivery system works: subcutaneous administration crosses the BBB in humans as it does in NHPs, opening the entire CNS target space to Arrowhead’s platform; and
(3) the platform re-rates: Arrowhead is no longer a cardio/metabolic RNAi company with a CNS program. It becomes a CNS-capable RNAi platform company competing for partnerships with every major pharma advancing into neurodegenerative disease.
The Novartis deal for ARO-SNCA (α-synuclein, Parkinson’s disease) at $200 million upfront closed in October 2025, months before the first ARO-MAPT dose. That deal was signed on the basis of preclinical data alone. ARO-MAPT clinical data would be worth multiples of that in any future partnership discussion.
The thesis in one sentence: ARO-MAPT is not an Alzheimer’s drug bet. It is a bet that Arrowhead has solved the hardest problem in RNA therapeutics, getting RNAi molecules into the brain after a subcutaneous injection, and that the market has not yet priced what that capability is worth.
II. Alzheimer’s Disease and the Tauopathy Spectrum
The Scope of the Problem
Alzheimer’s disease is the most common cause of dementia, affecting an estimated 32 million people worldwide and over 7 million Americans, a number projected to reach 13 million by 2050 as the population ages. It is the sixth leading cause of death in the United States. Total annual costs of care in the U.S. alone exceed $350 billion. No disease in medicine carries a larger combined burden of human suffering and economic cost for which approved treatments remain fundamentally inadequate.
Alzheimer’s is a progressive, irreversible neurodegenerative disorder characterized by two hallmark pathological features: extracellular amyloid-beta (Aβ) plaques and intraneuronal neurofibrillary tangles (NFTs) composed of hyperphosphorylated tau protein. The disease progresses through a preclinical phase (biomarker changes with no symptoms), mild cognitive impairment (MCI) due to Alzheimer’s, mild Alzheimer’s dementia, and eventually moderate to severe Alzheimer’s dementia.
Alzheimer’s Is Not the Only Tauopathy
The relevance of ARO-MAPT extends well beyond Alzheimer’s. Tau pathology is the defining feature of a spectrum of neurodegenerative diseases collectively called tauopathies. Each represents a distinct clinical and neuropathological entity, but all share the common mechanism of aberrant tau protein accumulation:
Frontotemporal Dementia (FTD), the second most common dementia in adults under 65; a significant subset is caused by MAPT mutations
Progressive Supranuclear Palsy (PSP), a relentlessly progressive disease with no approved disease-modifying treatment; tau tangles in specific brainstem nuclei drive the clinical picture
Corticobasal Degeneration (CBD), overlapping clinical and neuropathological features with PSP; tau aggregates in both neurons and glial cells
Chronic Traumatic Encephalopathy (CTE), the tau-driven consequence of repetitive head trauma; affects millions of contact sport athletes and military veterans
Pick’s Disease, a rare FTD variant defined by Pick bodies composed of 3-repeat tau isoforms
Collectively, the tauopathy spectrum represents a patient population well exceeding 40 million people globally. A subcutaneous RNAi therapeutic capable of reducing tau protein throughout the CNS would be relevant to all of them. The commercial logic of a single subcutaneous therapeutic platform addressing this entire spectrum is not incremental; it is transformational.
III. The Tau Hypothesis: Biology, Genetics, and the Therapeutic Case for MAPT Silencing
What Tau Does: Normal Function and Pathological Failure
The MAPT gene encodes microtubule-associated protein tau, a protein expressed predominantly in neurons whose primary physiological function is to stabilize microtubules, the structural scaffolding that maintains neuronal shape and facilitates axonal transport of essential cargo. In healthy neurons, tau is soluble, appropriately phosphorylated, and bound to microtubules. This is normal tau doing its job.
In tauopathies, this equilibrium fails. Tau becomes hyperphosphorylated at specific serine and threonine residues. Hyperphosphorylated tau dissociates from microtubules, becomes soluble in the cytoplasm, and begins to aggregate: first into oligomers, then into paired helical filaments, and ultimately into the large neurofibrillary tangles (NFTs) that are the neuropathological signature of Alzheimer’s and related diseases. The consequence is double-edged: loss of microtubule stability disrupts axonal transport (a gain-of-loss effect), and the aggregated tau species are themselves directly toxic to neurons (a gain-of-toxic-function effect).
Critically, the density of NFTs correlates directly with the severity of cognitive impairment in Alzheimer’s disease; a stronger correlation than amyloid plaque burden, as documented in multiple neuropathological studies and reviewed in Lleo et al. (Cell, 2025). This is not a contested observation. It is one of the most replicated findings in Alzheimer’s neuropathology.
The Genetic Argument for MAPT Reduction
The therapeutic rationale for reducing tau production via MAPT gene silencing is among the most genetically validated hypotheses in drug development. Multiple independent lines of evidence converge on the same conclusion: less tau is better.
MAPT mutations cause disease: Dominant mutations in MAPT cause familial FTD and PSP. The mutations do not eliminate tau function; they alter tau splicing or aggregation propensity. This establishes tau aggregation as the driver of pathology.
MAPT haploinsufficiency is benign: People and mice carrying only one functional copy of the MAPT gene are neurologically normal. Tau reduction is well-tolerated.
Trisomy 21 overexpresses tau: Individuals with Down syndrome, who carry three copies of chromosome 21 and thus have elevated levels of APP (amyloid precursor protein), develop AD-like pathology early in life. The downstream consequence includes elevated tau.
Mouse MAPT knockout is viable: Complete tau knockout mice develop and function normally under standard conditions, supporting that tau suppression does not produce catastrophic physiological consequences.
Antisense data: BIIB080 clinical data in Alzheimer’s patients have demonstrated that approximately 60% reduction in CSF tau is pharmacologically achievable and appears safe in early trials, providing direct human evidence that the mechanism is druggable.
The genetic case for MAPT silencing is not a hypothesis under debate. It is a hypothesis under execution. The question is not whether reducing tau protein is a valid therapeutic strategy. The question is whether Arrowhead’s RNAi approach can do it better than the competition.
Why Not Amyloid? The Tau Advantage
The approval of lecanemab (Leqembi) and donanemab (Kisunla) in 2023 validated the amyloid hypothesis and opened the era of disease modification in Alzheimer’s. But their commercial and clinical performance has been sobering. Efficacy is real but modest, 18-35% slowing of decline on composite clinical scales. Safety concerns, particularly amyloid-related imaging abnormalities (ARIA), complicate administration and require MRI monitoring, and the drugs are intravenous infusions administered in specialized centers.
More fundamentally, amyloid clearance does not stop neurodegeneration in already-affected neurons. The field’s growing consensus is that combination therapy combining anti-amyloid and anti-tau agents will be required for meaningful disease modification, analogous to multi-agent strategies in oncology and HIV. This is not a fringe view; it is the stated strategy of Biogen, Eli Lilly, Eisai, and multiple academic consortia. In this paradigm, an effective anti-tau agent does not compete with anti-amyloid therapies. It completes them.
IV. ARO-MAPT: Mechanism, Design, and the New CNS Delivery System
What ARO-MAPT Does
ARO-MAPT is an RNAi therapeutic designed to silence CNS expression of the MAPT gene. Its mechanism follows the canonical RNAi pathway: the therapeutic molecule, once delivered to neurons, is loaded into the RNA-induced silencing complex (RISC), which uses the antisense strand of the siRNA as a guide to identify and cleave MAPT mRNA. Cleaved mRNA is degraded, and tau protein production is suppressed. The catalytic nature of RISC is what enables deep, durable knockdown from infrequent dosing: one loaded RISC complex cleaves multiple mRNA molecules in succession, unlike ASOs which require stoichiometric hybridization.
ARO-MAPT targets all tau isoforms. The MAPT gene produces six main tau isoforms in the adult brain, arising from alternative splicing of exons 2, 3, and 10. Pathological aggregation involves specific isoform imbalances (e.g., 4-repeat tau predominates in PSP/CBD; 3-repeat in Pick’s disease; mixed isoforms in Alzheimer’s). By targeting a sequence common to all isoforms, ARO-MAPT has the potential to suppress total tau and, by reducing the total protein pool available for aggregation, address all tauopathies regardless of their specific isoform pathology.
The New CNS Delivery System: TfR1-Mediated BBB Penetration
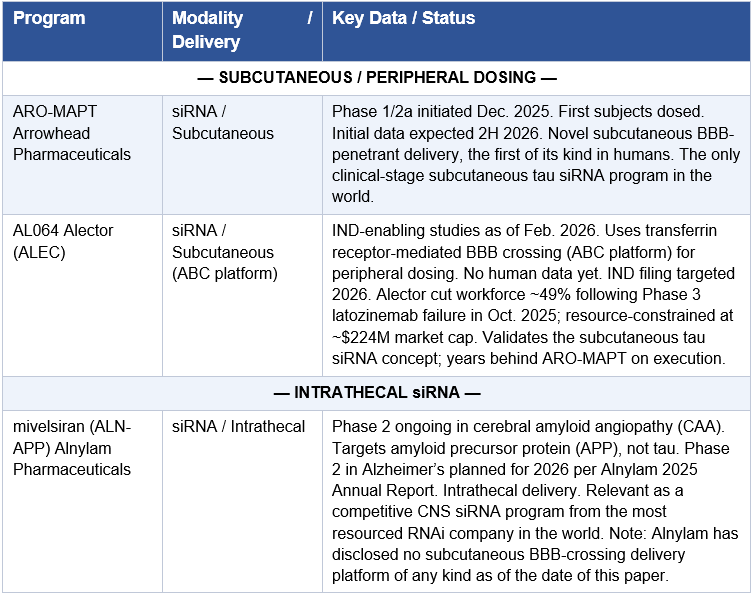
This is the single most important technical development in ARO-MAPT’s story. The history of RNAi therapeutics in the CNS has been defined by one word: intrathecal. Because GalNAc-conjugated siRNA does not penetrate the blood-brain barrier, every CNS RNAi and ASO program to date, from BIIB080 to mivelsiran to ALN-5288 to ALN-HTT02, has required intrathecal delivery: a lumbar puncture that places drug directly into the cerebrospinal fluid. Alnylam, the most technically advanced siRNA company in the world, has five CNS programs in their Alnylam 2030 five-year strategic plan. Every single one is delivered intrathecally or via IV. They have not disclosed a subcutaneous BBB-crossing delivery platform of any kind. The subcutaneous CNS delivery problem has not been solved by anyone, anywhere in the industry, until now.
Intrathecal delivery works pharmacologically. BIIB080 achieves approximately 56-60% tau reduction in human CSF. But intrathecal administration requires a trained clinician, a procedural suite, sedation in many patients, and risk of post-lumbar-puncture headache. A therapy requiring quarterly lumbar punctures will always be a specialty-center treatment, not a community care standard for a disease affecting over 7 million Americans.
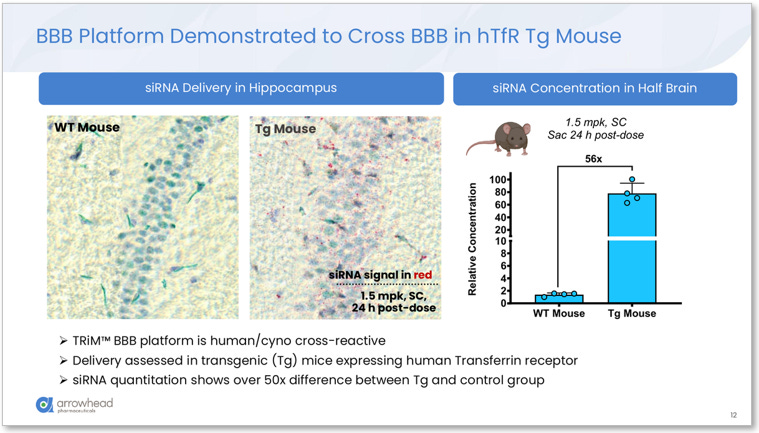
Arrowhead’s CNS delivery system resolves this. The TRiM™ BBB platform, presented in complete preclinical detail at the 7th Annual CNS Delivery Summit on December 10, 2025, by Dr. Agnieszka Glebocka, Vice President of Discovery Chemistry, works by conjugating the siRNA to a ligand targeting Transferrin Receptor 1 (TfR1). TfR1 is highly expressed on brain endothelial cells, and after subcutaneous injection the conjugate circulates systemically, engages TfR1 at the blood-brain barrier, undergoes receptor-mediated transcytosis into the brain parenchyma, and is taken up by neurons and other CNS cells. A cryo-EM structure presented in the slides confirms the ligand binds the apical domain of TfR1 and does not compete with endogenous transferrin binding. Iron transport is fully preserved, removing the primary theoretical safety concern with TfR1-targeting approaches.
Figure 1. TRiM™ BBB platform demonstrated to cross the BBB in hTfR transgenic mice expressing human TfR1. Left: hippocampal tissue staining showing dense siRNA signal (red) in Tg mice versus near-zero in wildtype controls at 1.5 mg/kg SC. Right: siRNA concentration in half-brain 56x higher in Tg mice than wildtype, confirming receptor-mediated transcytosis as the delivery mechanism. Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
ARO-MAPT NHP Data: The Full CNS Delivery Summit Dataset
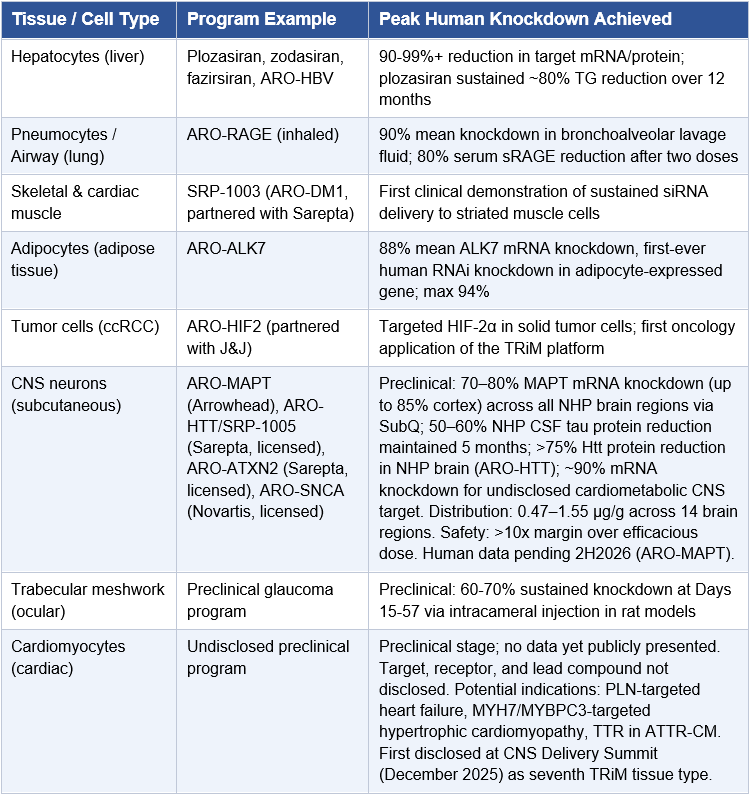
The December 10, 2025 slide deck contains the most comprehensive ARO-MAPT preclinical data Arrowhead has publicly released. The dataset covers four categories.
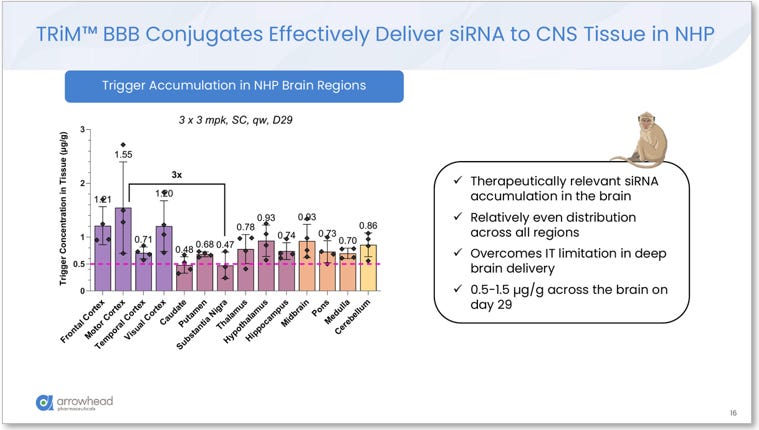
Delivery breadth: siRNA trigger accumulation was measured across 14 NHP brain regions after 3 x 3 mg/kg subcutaneous weekly doses, with NHPs sacrificed on Day 29. Values ranged from 0.47 μg/g (substantia nigra) to 1.55 μg/g (motor cortex), a ratio of approximately 3x across the entire brain. The slide explicitly states the distribution “Overcomes IT limitation in deep brain delivery.” For reference, the substantia nigra and pons, the structures most devastated in PSP, showed 0.47 μg/g and 0.73 μg/g respectively, within 3x of cortical concentrations. Intrathecal delivery cannot achieve this.
Figure 2. TRiM™ BBB siRNA trigger accumulation across 14 NHP brain regions (3 x 3 mg/kg SC, Day 29). Values range from 0.47 μg/g (substantia nigra) to 1.55 μg/g (motor cortex), a ~3x ratio demonstrating near-uniform vascular distribution. Arrowhead slide: “Overcomes IT limitation in deep brain delivery.” Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
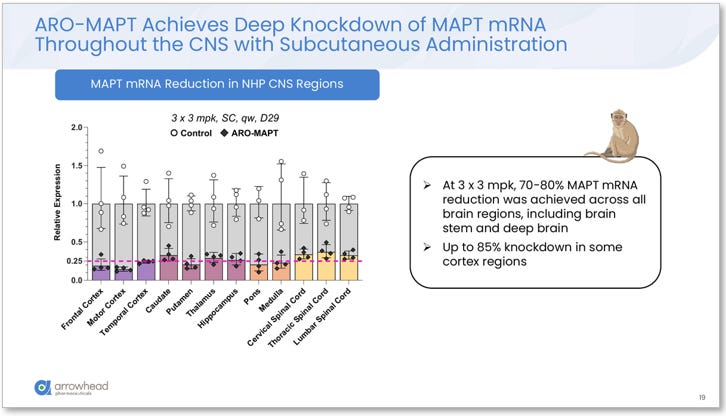
Knockdown depth (ARO-MAPT specific): At 3 x 3 mg/kg SC weekly (Day 29), ARO-MAPT achieved 70–80% MAPT mRNA reduction across ALL measured brain regions, including frontal cortex, motor cortex, temporal cortex, caudate, putamen, thalamus, hippocampus, pons, medulla, and cervical, thoracic, and lumbar spinal cord. Up to 85% knockdown was observed in some cortex regions. Hippocampal tissue staining corroborated the quantitative mRNA data, showing near-complete depletion of MAPT mRNA signal in treated versus control animals.
Figure 3. ARO-MAPT MAPT mRNA reduction across all measured NHP CNS regions (3 x 3 mg/kg SC, Day 29). Uniform 70–80% knockdown observed across frontal cortex, motor cortex, temporal cortex, caudate, putamen, thalamus, hippocampus, pons, medulla, and spinal cord. Up to 85% knockdown in some cortex regions. Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
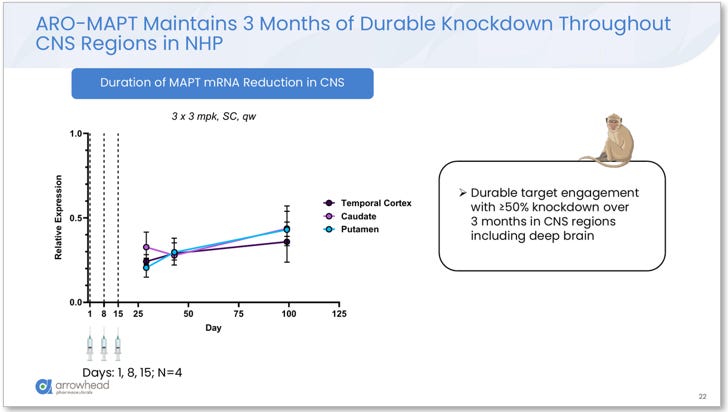
Figure 4. Durability of ARO-MAPT MAPT mRNA knockdown in NHP (3 x 3 mg/kg SC weekly loading doses). Greater than or equal to 50% knockdown maintained for over 3 months in temporal cortex, caudate, and putamen, consistent with monthly to quarterly dosing in humans. Source: Glebocka, 7th Annual CNS Delivery Summit, Dec. 10, 2025.
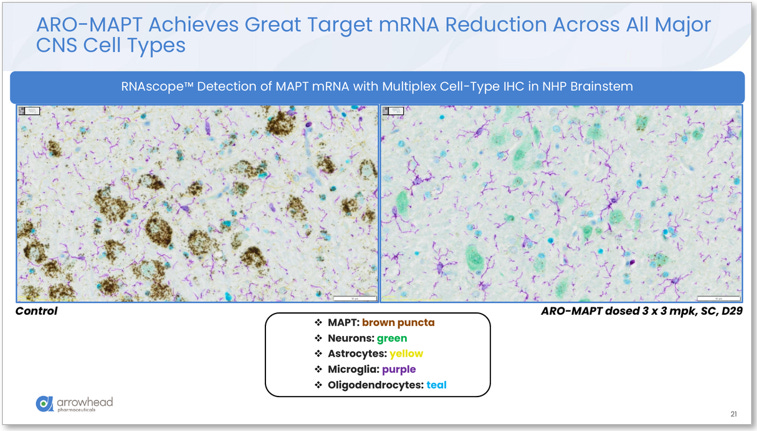
Cell type coverage: RNAscope multiplex immunohistochemistry in NHP brainstem confirmed MAPT mRNA knockdown across all four major CNS cell types: neurons, astrocytes, microglia, and oligodendrocytes. Tau pathology in tauopathies is not neuron-exclusive. Oligodendroglial tau inclusions are a hallmark of CBD; astrocytic tau pathology is prominent in multiple 4R tauopathies. Pan-cellular knockdown addresses this.
Figure 5. RNAscope™ multiplex IHC in NHP brainstem (3 x 3 mg/kg SC, Day 29). Left (control): abundant brown MAPT mRNA puncta across all cell types. Right (ARO-MAPT treated): near-complete MAPT mRNA depletion in neurons (green), astrocytes (yellow), microglia (purple), and oligodendrocytes (teal). Pan-cellular knockdown confirmed. Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
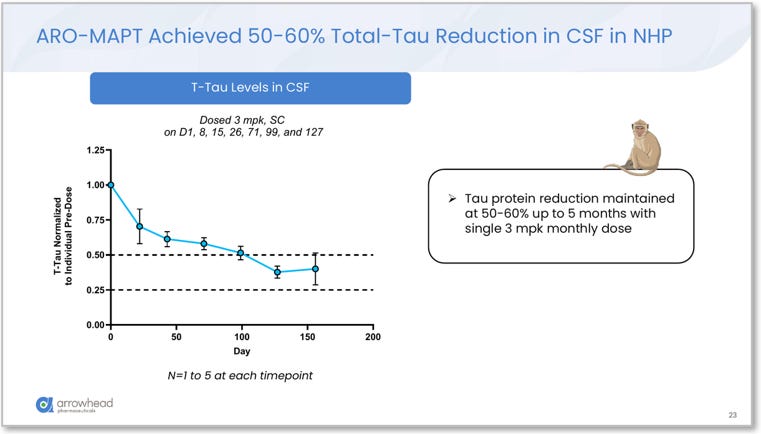
CSF tau protein: ARO-MAPT achieved 50–60% total-tau reduction in NHP CSF, maintained for up to 5 months with once-monthly 3 mg/kg subcutaneous dosing. This is the most analytically significant finding in the preclinical package and is addressed separately below.
Figure 6. ARO-MAPT NHP CSF total-tau reduction over time. Monthly subcutaneous dosing (3 mg/kg). 50–60% CSF total-tau reduction maintained for up to 5 months. This NHP result already falls in the same range as BIIB080’s Phase 1b human outcome of 56–60% CSF tau reduction via intrathecal injection. Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
ARO-MAPT achieved 70–80% MAPT mRNA knockdown across all NHP brain regions, with up to 85% in cortex, and then translated that mRNA reduction into 50–60% CSF total-tau protein reduction maintained for 5 months in NHPs. BIIB080 achieved 56–60% CSF tau reduction in humans via intrathecal injection. The ARO-MAPT NHP CSF tau data is already in the same range as BIIB080’s best human Phase 1b result. Via a subcutaneous injection.
The CSF Tau Comparison That Reframes the Entire Competitive Analysis
This point warrants its own analysis because it changes the entire benchmarking framework. The standard way investors and analysts have been thinking about ARO-MAPT is as follows: BIIB080 showed 56-60% CSF tau reduction in humans, and ARO-MAPT needs to demonstrate something comparable in its Phase 1/2a to validate the thesis. That framing is now incomplete.
The CNS Delivery Summit data shows ARO-MAPT has already demonstrated 50-60% CSF tau protein reduction in NHPs after subcutaneous dosing. This is not a prediction. It is a measured outcome. BIIB080’s 56-60% human CSF tau reduction was achieved after 13 weeks of monthly intrathecal injections. ARO-MAPT’s 50-60% NHP CSF tau reduction was achieved after a monthly subcutaneous dosing schedule and maintained for 5 months. The NHP CSF tau biomarker for ARO-MAPT is already in the same therapeutic range as the human result that sent BIIB080 into Phase 2.
The NHP-to-human translation step that is still required is whether ARO-MAPT’s vascular delivery mechanism works as effectively in humans as in cynomolgus monkeys. TfR1 expression on brain endothelial cells is conserved between cynomolgus macaques and humans, which is why the TRiM BBB platform was specifically designed using human/cyno cross-reactive chemistry. The platform is confirmed cross-reactive on slide 12 of the CNS Delivery Summit deck. This does not guarantee human replication, but it is the strongest possible preclinical foundation.
Cross-Platform Validation: ARO-HTT and the Undisclosed Target
The CNS Delivery Summit presentation also covered two additional TRiM BBB programs. ARO-HTT, the Huntington’s disease program partnered with Sarepta Therapeutics, achieved >75% Htt protein reduction throughout NHP brain regions at 3 x 3 mg/kg (frontal cortex, temporal cortex, caudate, putamen all confirmed), specifically targeting deep brain structures critical to HD pathogenesis. A second undisclosed cardiometabolic CNS target (“Target X” in the slides) achieved approximately 90% mRNA knockdown across all measured NHP brain regions at the same dose. The platform is not being optimized for one target in one disease. It is demonstrating consistent, deep, uniform CNS knockdown across three separate gene targets in NHP studies, with results ranging from 70-80% for MAPT to approximately 90% for the cardiometabolic target.
PK/PD Modeling and Projected Clinical Dosing
The CNS Delivery Summit deck presented a full PK/PD model derived from NHP tissue concentrations and tau mRNA data across 17 CNS tissues. Key projections: a monthly booster after the initial loading regimen projects to maintain approximately 80% MAPT mRNA knockdown; quarterly dosing projects to maintain 50–70% sustained knockdown. The model also included predicted human CNS concentrations, with the projected human pharmacokinetic profile confirming therapeutic exposure throughout the brain under the quarterly dosing scenario.
Safety and Formulation
Exploratory toxicology in both rodent and NHP showed a safety profile with greater than 10x margin over the efficacious dose, with no dose-limiting toxicities in either species. The ARO-MAPT formulation achieves 150 mg of siRNA in 4 mL or less total volume, confirming that therapeutically relevant doses can be delivered in a standard subcutaneous injection without IV-line administration or clinical observation requirements.
How the New Delivery System Fits Within TRiM
The TRiM™ (Targeted RNAi Molecule) platform is the connective tissue of Arrowhead’s entire pipeline. TRiM is not a single molecule; it is a modular design framework that combines a siRNA duplex with a targeting ligand engineered for cell-type-specific uptake. The liver-targeting TRiM molecules use GalNAc ligands that bind the asialoglycoprotein receptor (ASGPR) on hepatocytes. The muscle-targeting TRiM conjugates are designed for skeletal and cardiac muscle. The CNS-targeting molecules in ARO-MAPT use a TfR1-targeting ligand that achieves BBB penetration and neuronal uptake after systemic subcutaneous administration. Each tissue type required solving a distinct ligand-engineering problem: identifying a receptor with sufficient and selective expression on the target cell, designing a targeting moiety with appropriate binding kinetics, and optimizing the full TRiM conjugate for potency, stability, and tolerability.
The CNS Delivery Summit presentation confirmed that Arrowhead is actively advancing TRiM delivery platforms across seven distinct tissue types: liver (strong clinical validation), lung (deep lung clinical validation via RAGE), skeletal muscle (early clinical stage), CNS (early clinical stage, ARO-MAPT now enrolling), adipose (early clinical stage), ocular (preclinical), and cardiomyocyte (preclinical). The cardiomyocyte platform entry is notable because it was not on prior public-facing pipeline slides and represents the seventh tissue type Arrowhead has taken to some stage of TRiM development. CNS today, ocular and cardiomyocyte tomorrow. The platform keeps expanding.
Intellectual Property: The Patent Moat Around the BBB Platform
The competitive durability of Arrowhead’s TRiM BBB platform depends not only on the technical complexity of the delivery system but on the scope of its patent protection. Arrowhead has filed and been awarded multiple patents covering the TRiM platform architecture, including the targeting ligand design, the conjugation chemistry, and the siRNA optimization framework. The TfR1-targeting BBB delivery system specifically represents a distinct and novel approach to receptor-mediated transcytosis for oligonucleotide delivery: rather than using large protein-based ligands (antibody-receptor conjugates, as several competitors have attempted), the TRiM BBB conjugate uses a small-molecule-like ligand engineered for TfR1 binding with the pharmacokinetic stability required to circulate intact until it reaches the brain endothelium. This specific combination of a small-molecule TfR1 ligand, a stable non-reversible covalent linkage, and a potency-optimized siRNA duplex is the subject of patent coverage.
Competitors pursuing TfR1-mediated BBB delivery have generally relied on antibody-based or peptide-based receptor-targeting approaches, each with different binding kinetics, different immunogenicity profiles, and different manufacturability constraints. Alector’s ABC platform, the closest subcutaneous tau siRNA analog to ARO-MAPT, uses a different receptor-targeting approach. The specific molecular architecture of the TRiM BBB conjugate, its apical domain TfR1 binding confirmed by cryo-EM structure, its non-interference with endogenous transferrin, and its covalent stability in circulation, are not features that can be replicated without access to Arrowhead’s proprietary chemistry. For investors evaluating whether positive ARO-MAPT data creates a sustainable competitive position or merely a temporary head-start, the patent protection around the delivery mechanism is a meaningful moat. Arrowhead is not racing to be first in a technology that will be commoditized. It is building on a proprietary molecular architecture that competitors cannot simply reverse-engineer.
V. Clinical Program: AROMAPT-SC-1001
Trial Design Overview
The Phase 1/2a trial AROMAPT-SC-1001 (NCT07221344) was initiated on December 8, 2025, when Arrowhead dosed the first subjects. It is a placebo-controlled, dose-escalating study designed to evaluate the safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of ARO-MAPT. The trial has two parallel arms:
The trial is registered at ClinicalTrials.gov as AROMAPT-SC-1001 (NCT07221344). Arrowhead requested regulatory clearance on September 10, 2025 and announced first subject dosing on December 8, 2025 in a press release published the same day. The parallel structure of the trial is strategically important: running healthy volunteer and disease cohorts simultaneously accelerates the path to meaningful pharmacodynamic data without waiting for sequential Phase 1 completion. Initial data are expected in the second half of 2026.
What Data Will Be Reported in 2H 2026
The initial 2H 2026 readout will almost certainly focus on the healthy volunteer cohort, specifically safety, tolerability, and early PK/PD data. In this context, the most meaningful initial biomarker signal will be:
CSF Total Tau: The primary pharmacodynamic marker. Reduction from baseline in treated vs. placebo subjects is the primary efficacy signal.
CSF Phospho-Tau (p-tau181, p-tau217): Hyperphosphorylated tau species most directly implicated in tangle formation. Reduction here has the highest disease-relevance.
Plasma p-tau217: Increasingly validated as a blood-based proxy for CNS tau pathology. Less invasive than CSF sampling.
Neurofilament Light Chain (NfL): A marker of axonal injury and neurodegeneration. A neutral or declining NfL in treated subjects would suggest that tau reduction is not being accompanied by neuronal damage.
The 2H 2026 data must be interpreted against three benchmarks, not two. First, the BIIB080 Phase 1b human benchmark: 56–60% reduction in CSF total-tau in the highest-dose groups, achieved via intrathecal injection in human Alzheimer’s patients. Second, ARO-MAPT’s own NHP mRNA benchmark: 70–80% MAPT mRNA knockdown across all brain regions, up to 85% in cortex via subcutaneous injection, first presented at the 7th Annual CNS Delivery Summit (December 10, 2025). Third, and most importantly: ARO-MAPT’s own NHP CSF tau benchmark: 50–60% total-tau protein reduction in NHP CSF, maintained for up to 5 months with monthly subcutaneous dosing. This third benchmark is the most significant because CSF tau reduction is the exact biomarker that will be measured in human subjects in 2H 2026. BIIB080’s NHP MAPT mRNA knockdown (74–77%) translated to 56–60% human CSF tau reduction, a step-down. ARO-MAPT’s NHP CSF tau data is already 50–60%, which means in NHPs the two programs are already producing identical CSF tau outcomes. The human question is not whether ARO-MAPT will beat BIIB080 in CSF tau depth. The question is whether ARO-MAPT’s NHP CSF result translates to humans at all. A human CSF tau reduction of 40–60% with a clean safety profile would be a positive readout. A result at or above 60% would establish ARO-MAPT as the most potent anti-tau agent in the clinical landscape.
The Sub-Q Delivery Advantage in Clinical Context
It is worth pausing on what subcutaneous delivery means in the context of a disease as prevalent as Alzheimer’s. The patients who need anti-tau therapy are not young, healthy adults. They are elderly patients, mean age 70-80 in most AD trials, many of whom have mobility limitations, comorbidities, and caregivers who manage their medications. The prospect of a lumbar puncture every 3-4 months for the rest of their lives is a significant barrier to treatment initiation and adherence. Subcutaneous injection, in contrast, can potentially be self-administered at home or by a caregiver, analogous to insulin or GLP-1 therapies.
This is not a trivial commercial distinction. The entire lesson of the GLP-1 revolution (explored in a prior BioBoyScout white paper, The Needle Wins, on subcutaneous RNAi delivery and commercial scalability) is that the modality of delivery shapes the commercial outcome as much as the pharmacology. An anti-tau therapy that can be administered subcutaneously at home, once a month, will capture a meaningfully larger share of the treatable patient population than one requiring clinic visits for lumbar punctures.
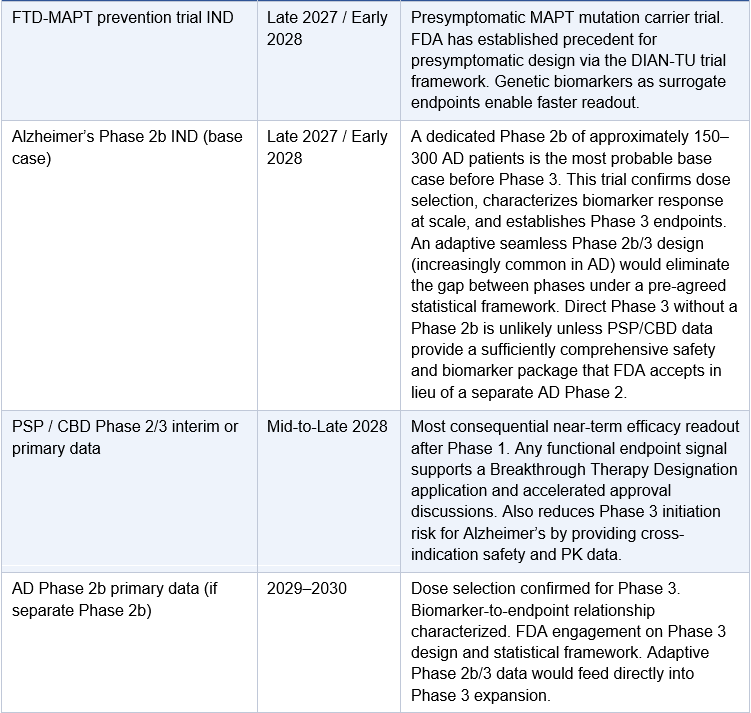
From Phase 1 to First Approval: The Regulatory Roadmap
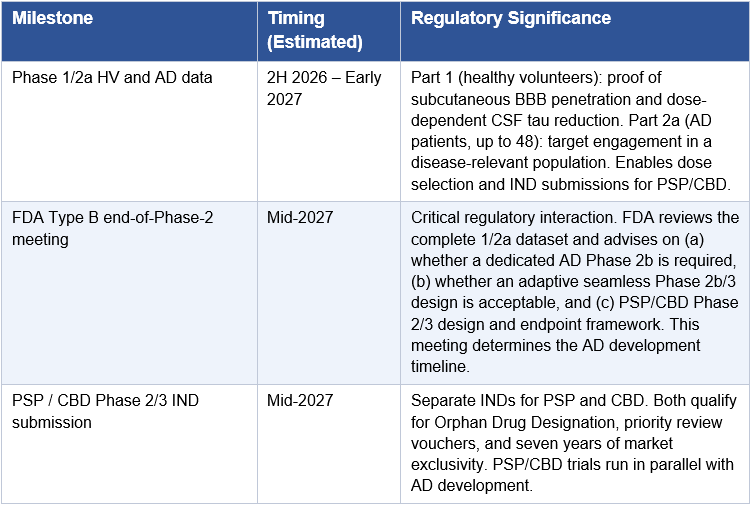
A successful 2H 2026 Phase 1 readout does not immediately translate to an approved drug. Understanding the operational sequence from Phase 1 data to first commercial approval is essential for investors modeling the timeline. The following represents Arrowhead’s most probable regulatory pathway assuming the Phase 1 data confirms subcutaneous BBB penetration and meaningful tau reduction.
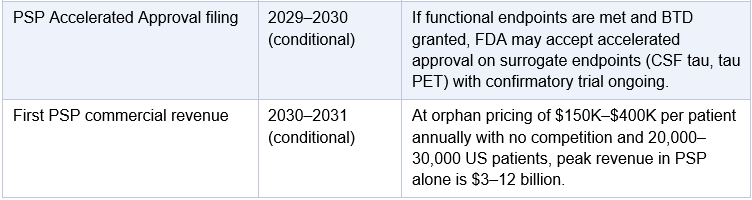
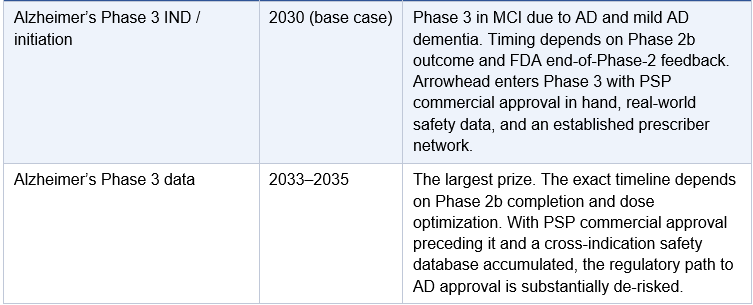
One point requires specific clarification: Arrowhead will almost certainly not proceed directly from the Phase 1/2a to a Phase 3 trial in Alzheimer’s disease. The AROMAPT-SC-1001 Part 2a AD cohort enrolls up to 48 patients in a dose-escalating design. That is a Phase 2a: it establishes target engagement and safety in AD patients but does not provide the dose-response characterization, biomarker durability, or statistical power FDA requires before sanctioning a 1,500–2,000 patient registrational trial for a novel delivery mechanism with no prior human CNS precedent. Every approved Alzheimer’s therapy, including lecanemab and donanemab, followed a full Phase 1, Phase 2b, Phase 3 sequence. BIIB080 ran a 46-patient Phase 1b followed by a 416-patient Phase 2 before any Phase 3 was planned. The base case for ARO-MAPT is a dedicated AD Phase 2b of approximately 150–300 patients running approximately 12–18 months, following the Phase 1/2a dataset. The exception would be an adaptive seamless Phase 2b/3 design, in which a pre-agreed statistical framework allows Phase 2b dose-finding to roll directly into Phase 3 expansion without a separate IND or protocol restart. FDA has been receptive to this design in AD under Breakthrough Therapy Designation. If Arrowhead obtains BTD after Phase 1 data and agrees an adaptive design at the end-of-Phase-2 meeting, the gap between Phase 2b and Phase 3 collapses. This is the most optimistic realistic scenario; it is not the base case, but it is achievable if the Phase 1 data is strong and the PSP/CBD cross-indication safety database is sufficiently mature by mid-2027.
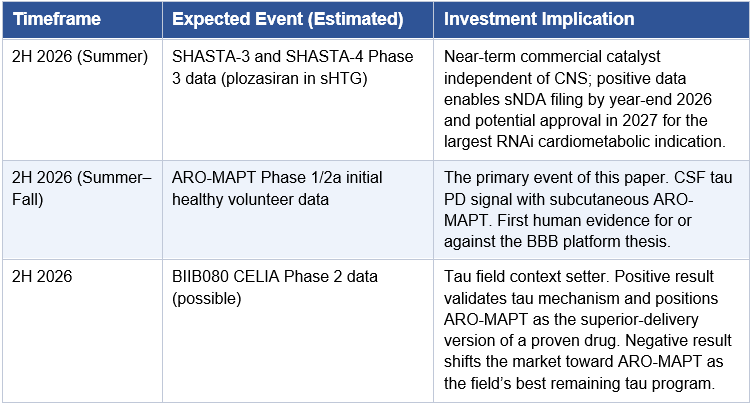
The Catalyst Calendar: 2026 to 2029
The following calendar consolidates all major expected Arrowhead events across the ARO-MAPT program and the broader pipeline. It is presented to give investors a sequenced view of the newsflow that will drive valuation over the next four years.
VI. Competitive Landscape: The Tau Space in 2026
The Tau Space in 2026: A Field at Inflection
Multiple industry analysts and key opinion leaders have described 2026 as ‘the year of tau’ in neurodegeneration drug development. Several major data readouts are expected across the tau-targeting competitive landscape, including efficacy data from BIIB080’s Phase 2 CELIA trial. Arrowhead enters this landscape at precisely the right moment, with a clinically differentiated approach on delivery modality. Understanding the competitive field is essential to contextualizing what a positive ARO-MAPT result would mean.
The competitive table below is organized to reflect the single most consequential variable in the tau space: delivery route. Programs are grouped by whether they can be administered peripherally or require CNS access via lumbar puncture or IV infusion. That distinction will determine which therapies can scale to the tens of millions of patients who need them.
The ASO vs. RNAi Question in the CNS
BIIB080 is the gold standard against which all tau-targeting programs, including ARO-MAPT, will be evaluated. BIIB080 is an antisense oligonucleotide: a single-stranded DNA-like molecule that binds tau mRNA and recruits RNase H to degrade it. Like all current CNS ASOs, it must be delivered intrathecally. Ionis has been the dominant force in CNS ASO development for two decades. Biogen paid $1.5 billion in upfront licensing to gain access to BIIB080, a testament to the commercial value Biogen ascribed to validated tau knockdown. The Phase 1b peer-reviewed data (PubMed) showed dose-dependent, sustained reductions in CSF tau across all dose groups by the end of the long-term extension, with the two highest-dose cohorts achieving 56% reduction in CSF total-tau and 51% reduction in p-tau181 at the end of the long-term extension. Across all high-dose cohorts, CSF total-tau reduction ranged from 56–60%, as detailed in NeurologyLive’s clinical coverage. The Phase 2 CELIA baseline characteristics have been published in full, enrolling 416 participants aged 50-80 with MCI or mild AD dementia, confirming the robust Phase 2 infrastructure behind this program. In April 2025, BIIB080 received FDA Fast Track Designation for Alzheimer’s disease, further validating the tau-reduction mechanism.
RNAi (siRNA) and ASOs share a common goal, reducing target mRNA, but differ fundamentally in mechanism and molecular pharmacology. The catalytic nature of RISC is what enables deep, durable knockdown from infrequent dosing: one loaded RISC complex cleaves multiple mRNA molecules in succession, unlike ASOs which require stoichiometric hybridization. In liver programs, Arrowhead has consistently demonstrated that quarterly or semi-annual siRNA dosing achieves equivalent or superior knockdown compared to monthly ASO dosing. The NHP head-to-head is now quantified for the CNS: BIIB080 achieved 74–77% MAPT mRNA reduction via intrathecal delivery in cynomolgus monkeys; ARO-MAPT achieved 70–80% MAPT mRNA reduction across all brain regions (up to 85% in cortex) via subcutaneous injection, first presented at the 7th Annual CNS Delivery Summit on December 10, 2025. More importantly, ARO-MAPT then translated that mRNA knockdown into 50–60% NHP CSF tau protein reduction maintained for 5 months: the exact same range as BIIB080’s best human Phase 1b result, achieved via lumbar puncture.
Deep Brain Distribution: The Structural Advantage That Phase 1 Biomarkers Cannot Fully Reveal
The single most underappreciated dimension of the ARO-MAPT versus BIIB080 comparison is not headline knockdown depth. It is the geographic distribution of that knockdown throughout the brain. Understanding this distinction requires understanding how each drug actually reaches neurons, and what the physics of that delivery mean for the regions where tau pathology does its worst damage.
Why Intrathecal Delivery Creates a Concentration Gradient
BIIB080 is injected via lumbar puncture into the cerebrospinal fluid. The drug must then travel from the lumbar injection site upward through the spinal cord, into the subarachnoid space surrounding the brain, and from there diffuse across the pia mater into the brain parenchyma. This journey is governed by CSF fluid dynamics, not by active targeting. The result is an unavoidable pharmacokinetic gradient: structures adjacent to the CSF interfaces, particularly the periventricular regions, frontal cortex, and hippocampus, receive the highest drug concentrations. Structures deep in the brain parenchyma, far from any CSF-contacting surface, receive progressively less. This is not a design flaw unique to BIIB080. It is a physical constraint that applies to every intrathecally delivered CNS therapeutic, regardless of modality.
BIIB080’s own published NHP data documented this gradient: 77% MAPT mRNA knockdown in frontal cortex and 74% in hippocampus, the two regions most accessible to CSF diffusion. No peer-reviewed publication has reported BIIB080 NHP knockdown data for the substantia nigra, the subthalamic nucleus, the red nucleus, the dentate nucleus of the cerebellum, or the rostral interstitial nucleus of the medial longitudinal fasciculus. These are not obscure structures. They are, collectively, the anatomical locus of PSP. Their absence from the published BIIB080 NHP dataset is not an oversight; it is a reflection of the difficulty of demonstrating adequate drug exposure in regions that intrathecal delivery reaches least reliably.
What ARO-MAPT’s Distribution Data Shows
ARO-MAPT distributes via the vasculature. TfR1 is expressed on brain endothelial cells throughout the entire cerebrovascular tree. After subcutaneous injection, the TfR1-conjugated siRNA circulates systemically, engages TfR1 at the blood-brain barrier simultaneously across every vascularized structure in the brain, crosses by receptor-mediated transcytosis, and is taken up by neurons and other CNS cells throughout the parenchyma. There is no CSF gradient because there is no CSF. The drug arrives at every brain region through the same bloodstream at the same time.
The CNS Delivery Summit data quantified this distribution across 14 brain regions. Values ranged from 0.47 μg/g in the substantia nigra to 1.55 μg/g in the motor cortex: a ratio of approximately 3x between the highest and lowest region. In drug distribution terms, 3x variation across 14 brain regions is essentially uniform, particularly when the lowest value still sits above the threshold required for productive RISC loading. The resulting MAPT mRNA knockdown was 70–80% across every measured region, including the pons (0.73 μg/g), the medulla (0.70 μg/g), and the thalamus (0.78 μg/g). All of these structures are distal territory for intrathecal delivery. For ARO-MAPT, they are no different from cortex.
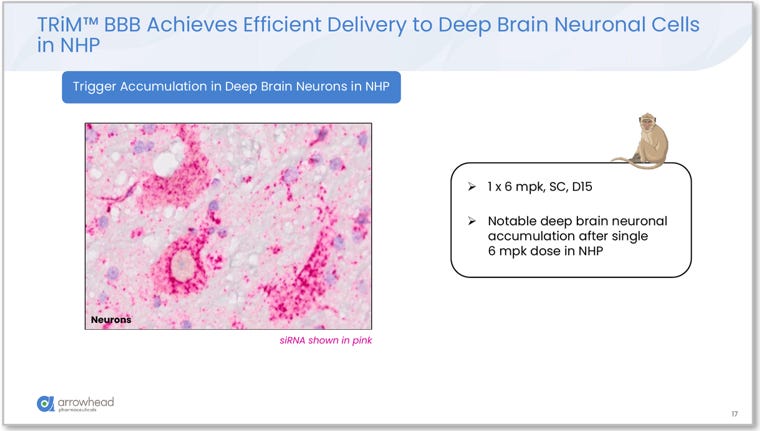
Figure 7. siRNA accumulation in deep brain neuronal cells in NHP (1 x 6 mg/kg SC, Day 15). Pink signal (siRNA) visible within deep brain neurons after a single subcutaneous dose, confirming vascular delivery reaches structures that intrathecal administration cannot adequately access. Source: Glebocka, 7th Annual CNS Delivery Summit, 12/10/2025.
The CSF Tau Biomarker as an Incomplete Readout
There is a critical analytical subtlety that investors must understand when interpreting the 2H 2026 Phase 1 data: CSF total-tau is a biased biomarker that disproportionately reflects tau production from cortical and hippocampal neurons. These regions have the highest neuronal density, the most direct CSF interface, and contribute the most to the lumbar CSF tau pool that is actually sampled during a lumbar puncture. Brainstem and deep subcortical contributions to lumbar CSF tau are real but diluted by the time they reach the sampling site.
The practical consequence is that ARO-MAPT and BIIB080 may show similar CSF tau reductions in Phase 1 human data while ARO-MAPT simultaneously achieves substantially more complete tau knockdown in the substantia nigra, subthalamic nucleus, midbrain, and brainstem. That deeper activity will not be fully captured in the lumbar CSF tau readout. It will be invisible in the Phase 1 headline numbers but will become clinically decisive in Phase 2 and Phase 3 trials for PSP and CBD, where those structures are the primary sites of pathology and where functional endpoints such as gaze velocity, balance, and motor control are driven by their integrity.
Do not read a similar Phase 1 CSF tau reduction as evidence that ARO-MAPT and BIIB080 are equivalent. They are measuring overlapping but not identical things. BIIB080’s CSF tau reduction largely reflects cortical and hippocampal activity. ARO-MAPT’s reduction reflects cortical, hippocampal, and deep brain activity, with the deep brain contribution partially masked by the anatomy of the sampling methodology.
The Cell Type Dimension: Pan-Cellular vs. Neuron-Predominant Knockdown
A further dimension of the distribution advantage bears directly on CBD and other glial tauopathies. ARO-MAPT’s RNAscope multiplex immunohistochemistry data from the CNS Delivery Summit showed MAPT mRNA knockdown confirmed across all four major CNS cell types in the NHP brainstem: neurons, astrocytes, microglia, and oligodendrocytes. This pan-cellular knockdown is a direct consequence of vascular delivery, since TfR1 is expressed on the endothelial cells that supply all cell types, and once the drug crosses into brain parenchyma it is available for uptake by any cell that engages the downstream internalization machinery.
CBD is characterized not only by neuronal tau inclusions but by astrocytic plaques and coiled bodies, which are tau-containing oligodendroglial inclusions prominent in both CBD and PSP. A drug that silences MAPT in neurons while leaving astrocytic and oligodendroglial MAPT expression intact addresses part of the pathological substrate, not all of it. BIIB080’s intrathecal ASO delivery and its glial uptake profile in deep brain structures are not characterized in the published literature. ARO-MAPT’s vascular delivery with confirmed pan-cellular knockdown in the brainstem closes that gap with published data.
The Phase 1 CSF tau biomarker data for ARO-MAPT and BIIB080 will likely look similar. They are not equivalent. BIIB080’s reduction reflects knockdown primarily in cortex and hippocampus. ARO-MAPT’s reduction reflects knockdown throughout the entire brain including the deep structures that drive PSP and CBD, with contributions from neurons, astrocytes, microglia, and oligodendrocytes. The biomarker does not capture this distinction. The Phase 2 and Phase 3 clinical outcomes will.
The Subcutaneous Divide: Who Has Solved the Delivery Problem
The most important analytical lens for reading this competitive table is simple: who has solved subcutaneous CNS delivery, and who hasn’t. Every intrathecal or IV program in the tau space, no matter how well-funded or how strong the mechanistic data, carries a ceiling on its commercial and clinical addressability. Arrowhead and Alector are the only two companies pursuing subcutaneous tau siRNA. Of those two, only Arrowhead has dosed humans.
Alector’s AL064 (ABC-enabled tau siRNA) uses transferrin receptor-mediated BBB crossing rather than Arrowhead’s ligand-receptor TRiM delivery system. The ABC platform has demonstrated preclinical brain penetration but has not yet filed an IND. Alector’s organizational situation compounds the execution risk: a ~49% workforce reduction following their Phase 3 latozinemab failure in October 2025 left the company with a $224M market cap and a sharply reduced development team. AL064 validates the subcutaneous tau siRNA concept; it does not threaten ARO-MAPT’s first-mover position in humans.
The most strategically significant entry in the intrathecal column is Eli Lilly’s LY-3954068. Lilly is the dominant Alzheimer’s company: it markets Kisunla (donanemab), the leading anti-amyloid therapy, and is investing aggressively in combination approaches. The fact that Lilly chose to advance a tau siRNA via intrathecal delivery rather than subcutaneous is not a statement about their preference for IT. It is a statement about the state of the technology: subcutaneous CNS siRNA delivery was not available to them when they made that decision. If ARO-MAPT validates subcutaneous delivery in humans, Lilly is exactly the kind of acquirer or partner who would move swiftly to access that capability, whether through a licensing deal or a full acquisition. Their existing tau siRNA investment deepens rather than reduces their incentive to acquire a subcutaneous solution.
Alnylam’s position in the CNS space deserves specific analysis because it is the most analytically significant data point in the competitive landscape. Alnylam’s 2025 Annual Report, released on April 7, 2026, just one day before this paper was published, reveals a five-year Alnylam 2030 strategic plan with five CNS programs across Huntington’s disease (ALN-HTT02), Alzheimer’s disease (mivelsiran targeting APP, and ALN-5288 targeting MAPT), ALS (ALN-SOD), and Parkinson’s disease (ALN-SNCA). Every single one uses intrathecal or IV delivery. Alnylam has publicly disclosed no subcutaneous BBB-crossing delivery technology of any kind. This is not a gap Alnylam is unaware of or indifferent to. It is a gap that reflects the genuine technical difficulty of receptor-mediated transcytosis across the blood-brain barrier after subcutaneous administration. The fact that the most well-funded, most technically sophisticated siRNA company in the world, with $2.9 billion in cash and over two decades of siRNA chemistry expertise, has not publicly disclosed a subcutaneous CNS delivery solution is the strongest possible independent confirmation that Arrowhead has solved something genuinely hard. If ARO-MAPT succeeds, the pressure on Alnylam to either develop their own subcutaneous CNS capability or access Arrowhead’s would be immediate and intense. ALN-5288, their MAPT-targeting tau siRNA, would be the most directly affected program: it is pharmacologically equivalent to ARO-MAPT in mechanism, and if ARO-MAPT validates subcutaneous delivery, ALN-5288 becomes the inferior-modality version of the same drug.
Voyager Therapeutics’ VY1706 represents a distinct approach: it packages a tau siRNA payload inside an IV-delivered AAV capsid engineered to cross the BBB. Preclinical NHP data showed 44-73% tau mRNA knockdown from a single IV dose, with broad brain distribution. The one-time or infrequent dosing potential is compelling, but IV delivery, AAV immunogenicity risks, and the regulatory complexity of gene therapy are meaningful headwinds. VY1706 is still in IND-enabling studies. It is more a long-term option than a near-term competitive threat to ARO-MAPT, but its existence further confirms that tau silencing via gene delivery to the brain is considered by multiple serious players to be one of the highest-value targets in neurodegeneration.
The Antibody Programs: A Different Mechanism, Similar Outcome
The anti-tau antibody programs (semorinemab, bepranemab, JNJ-63733657, PRX005) represent a fundamentally different approach: rather than preventing tau production, they target extracellular or soluble tau protein for clearance. Results have been mixed. Semorinemab’s LAURIET trial failed to show significant slowing in moderate AD. Bepranemab’s data have been more encouraging in early cohorts. The antibody approach faces the inherent challenge of targeting intracellular aggregates from the extracellular space. Reduction of tau production via gene silencing addresses the problem upstream: prevent the substrate from accumulating in the first place.
ARO-MAPT vs. BIIB080: The Head-to-Head Summary
The following table consolidates every key dimension of the ARO-MAPT and BIIB080 comparison into a single reference. Given how frequently this comparison will be made as 2H 2026 data approaches, this exhibit is intended as a navigational anchor for the analysis distributed across this paper.
The Broader Implication: What Validated Subcutaneous CNS Delivery Means for the Entire Intrathecal Field
The competitive table above lists seven intrathecal CNS gene-silencing programs across multiple companies and targets. Every single one of them was developed on the same foundational assumption: that subcutaneous CNS RNAi delivery is not possible, and therefore that lumbar puncture is the only viable route for getting siRNA or ASOs into the brain. That assumption has shaped every pipeline decision, every clinical trial design, every partnership structure, and every analyst valuation model in CNS gene silencing for the past decade.
ARO-MAPT is a direct challenge to that assumption. If the 2H 2026 Phase 1 data confirms subcutaneous BBB penetration and meaningful tau reduction in humans, the implications extend well beyond a single tau program. They call into question the strategic rationale for every currently planned intrathecal CNS gene silencing program that has not yet been dosed in humans.
To be precise: validated subcutaneous CNS delivery does not immediately render already-approved intrathecal drugs obsolete. Tofersen for SOD1 ALS continues treating patients. BIIB080, if it ultimately receives approval, will serve the patients already enrolled in its clinical infrastructure. Drugs in late-stage trials with enrolled patients will complete those trials. The obsolescence argument is not about overnight replacement. It is about what happens to new program development, physician choice, patient preference, and partner economics from the moment subcutaneous delivery is validated forward.
Consider the physician choice argument first. When a physician has a choice between a drug requiring a lumbar puncture every three to four months for the rest of a patient’s life, and a drug achieving equivalent or superior CNS gene silencing via a subcutaneous injection the patient can receive at home or in a primary care office, that physician will almost universally choose the subcutaneous version. Not because the intrathecal version fails, but because the subcutaneous version is better in every dimension that matters to patients with progressive neurodegenerative disease: comfort, adherence, scalability, accessibility, and the dignity of not requiring a clinical procedure every quarter. The commercial consequences of physician and patient preference are not subtle. They are the difference between a drug that reaches its addressable patient population and one that does not.
The partner economics argument is equally decisive. A large pharma company considering whether to license or develop a new CNS gene silencing program after ARO-MAPT human validation would face a binary choice: build on a subcutaneous platform with validated human BBB penetration, or build on an intrathecal platform that requires lumbar puncture and carries the ceiling that entails. No rational drug development organization, given that choice, selects the intrathecal path for a new program. The addressable market for a subcutaneous version is structurally larger. The peak revenue ceiling is higher. The reimbursement logic is cleaner. The patient recruitment for clinical trials is faster. Every economic argument points in the same direction.
This logic applies with particular force to Alnylam, and the timing of this paper makes the point sharper than it has ever been. On April 7, 2026, Alnylam published their 2025 Annual Report, which introduces the Alnylam 2030 strategy: a formal, board-approved, five-year strategic plan outlining where the company intends to invest, what it intends to build, and how it intends to compete through the end of the decade. The neuroscience pipeline in that plan includes five CNS programs: mivelsiran (ALN-APP) for cerebral amyloid angiopathy and Alzheimer’s, ALN-5288 for tau-targeted Alzheimer’s, ALN-HTT02 for Huntington’s disease, ALN-SOD for ALS, and ALN-SNCA for Parkinson’s disease. Every single one is delivered intrathecally or via IV. The Alnylam 2030 five-year plan contains no mention of subcutaneous CNS delivery, no disclosure of a BBB-crossing delivery platform, and no indication that subcutaneous CNS administration is a goal, a program, or even a research priority for the next five years. This is not a casual omission. A five-year strategic plan published by a $42 billion company with $2.9 billion in cash, 25 years of siRNA chemistry expertise, and a board mandate to achieve CNS leadership, does not accidentally leave out a subcutaneous CNS delivery platform. If Alnylam had one, or believed they would have one in the next five years, it would be in this document. It is not. The ramifications for Arrowhead are significant. Alnylam’s 2030 plan is, in effect, an independent confirmation that subcutaneous CNS RNAi delivery remains an unsolved problem for the world’s most capable siRNA company. If ARO-MAPT solves it in humans in 2H 2026, Arrowhead owns a capability that the most well-resourced company in the field has explicitly not committed to building, and the five CNS programs in Alnylam’s own 2030 plan become the clearest possible illustration of what an intrathecal strategy looks like in a world where a subcutaneous alternative exists.
If ARO-MAPT validates subcutaneous BBB penetration in humans, the investment thesis is not simply that ARO-MAPT is a better tau drug than BIIB080. It is that Arrowhead has solved a delivery problem that the most well-resourced company in the RNAi industry has not solved, and that this solution structurally disadvantages every current and future intrathecal CNS gene silencing program when physicians, patients, partners, and payors are making choices. The seven intrathecal programs in the competitive table above were designed before subcutaneous CNS RNAi was possible. The programs designed after it is validated will look different, and Arrowhead will be the reason why.
The CELIA Timing Question: What Happens If BIIB080 Reports First
BIIB080’s Phase 2 CELIA trial enrolled its 416th participant in April 2025 and is expected to report primary endpoint data in 2026. The timing of that readout relative to ARO-MAPT’s 2H 2026 Phase 1 data is not precisely known, and the order of reporting carries meaningfully different implications for how the market will frame ARO-MAPT’s data.
If CELIA reports before ARO-MAPT’s Phase 1 data, two scenarios apply. A positive CELIA result, meaning BIIB080 demonstrably slows cognitive or functional decline in Alzheimer’s patients, validates the tau-reduction therapeutic hypothesis with Phase 2 efficacy data from the most advanced program in the field. In this scenario, ARO-MAPT’s subsequent Phase 1 data would be received as the first human demonstration of a subcutaneous version of a drug whose Phase 2 clinical benefit has already been established. Every investor watching the tau space would understand immediately that the delivery modality is the remaining variable. A negative CELIA result carries more nuance. It does not invalidate tau reduction as a therapeutic strategy; it may instead indicate that tau reduction alone, delivered intrathecally with its distributional limitations, is insufficient for meaningful clinical benefit in a mixed Alzheimer’s population with variable amyloid co-pathology. In that scenario, ARO-MAPT’s deep brain distribution and subcutaneous administration become the proposed solution to BIIB080’s demonstrated limitation, not a program tainted by BIIB080’s failure.
If ARO-MAPT’s Phase 1 data reports before CELIA, the framing reverses. ARO-MAPT would enter the public data landscape as the first subcutaneous tau-lowering program to demonstrate human BBB penetration and CSF tau reduction. Any investor modeling the tau space would face CELIA data knowing that a pharmacologically equivalent or superior program via subcutaneous delivery already exists in Phase 1. A positive CELIA result in that context would be read as validating the mechanism underlying a drug that already has a better-delivery version in development. The CELIA outcome, whenever it arrives, is analytically constructive for ARO-MAPT in both timing scenarios. The key risk would be a negative CELIA result reported before ARO-MAPT Phase 1 data, which could create a short-term guilt-by-association selling event even though the structural arguments for ARO-MAPT remain intact.
Either CELIA outcome is ultimately constructive for ARO-MAPT. A positive result validates the mechanism. A negative result, if it reflects intrathecal delivery limitations rather than target biology, frames subcutaneous delivery as the solution. The only scenario that would genuinely impair ARO-MAPT is a negative CELIA result accompanied by scientific consensus that tau reduction at any depth via any delivery route cannot slow Alzheimer’s progression. The current body of evidence makes that scenario unlikely, given the NFT-cognition correlation and the genetic evidence for MAPT as a disease driver.
VII. The Novartis / ARO-SNCA Precedent: What the Market Already Paid for CNS RNAi
The Deal That Prices the Platform
On October 17, 2025, Arrowhead closed a global licensing and collaboration agreement with Novartis for ARO-SNCA, its investigational siRNA therapy targeting alpha-synuclein (α-syn) for the treatment of synucleinopathies, primarily Parkinson’s disease. Under the terms of the original agreement (announced September 2, 2025), Novartis paid $200 million upfront and is eligible to receive up to $2 billion in milestone payments plus royalties. The structure mirrors what Arrowhead would likely seek for ARO-MAPT partnership terms, at a higher starting point, given human clinical data.
This deal deserves careful analysis in the context of ARO-MAPT. ARO-SNCA is preclinical. It has not been dosed in humans. Novartis paid $200 million for rights to an investigational RNAi program against a CNS target using the same novel subcutaneous CNS delivery system being tested in ARO-MAPT. In other words, Novartis underwritten the hypothesis that Arrowhead’s subcutaneous CNS delivery works, before a single human has received it.
The deal was not just about α-synuclein. It included rights to collaborate on additional targets utilizing the TRiM CNS platform. Novartis, one of the world’s largest pharmaceutical companies with an enormous neurology franchise, is making a strategic bet that Arrowhead’s subcutaneous CNS delivery technology represents a platform, not a one-time innovation.
What Positive ARO-MAPT Human Data Would Do to Partnership Valuations
If ARO-SNCA’s preclinical CNS delivery data was worth $200 million in upfront licensing fees, then clinical data demonstrating human tau knockdown via subcutaneous administration would be worth multiples of that. Here is the logic:
Risk reduction: Arrowhead has disclosed 70–80% MAPT mRNA knockdown across all NHP brain regions (up to 85% in cortex) AND 50–60% NHP CSF tau protein reduction maintained for 5 months via subcutaneous dosing. Moving from that preclinical benchmark to confirmed human delivery data eliminates the single largest risk in any CNS platform licensing deal: the question of whether the delivery system works in humans. That de-risking step is worth enormous premium in licensing valuations; it was the precise risk that Novartis was paying $200 million to take in the ARO-SNCA deal.
Platform validation: Positive ARO-MAPT data validates the same delivery technology that underpins ARO-SNCA, ARO-MAPT itself, and potentially dozens of future CNS programs. The value does not stay in one program.
Competitive dynamic: As the only company with a clinically validated subcutaneous CNS RNAi delivery system, Arrowhead would become the go-to partner for any large pharma looking to develop CNS RNAi. The competitive moat deepens.
Pricing power: Arrowhead negotiated from a position of preclinical data for $200M upfront. From a position of clinical data showing human tau knockdown, every future deal would be bid more aggressively.
Novartis paid $200 million upfront for preclinical CNS delivery data. ARO-MAPT has now disclosed greater than 75% MAPT mRNA knockdown in NHP CNS tissue via subcutaneous injection at clinically translatable doses, the same delivery technology, now further advanced and disclosed in specific quantitative terms. Human clinical data is the next value inflection, and it is not yet priced into the stock.
VIII. The Market Opportunity: Sizing the Tau Prize
Alzheimer’s Disease Alone
The global Alzheimer’s disease market is expected to exceed $15 billion annually by the early 2030s based on current anti-amyloid therapy uptake and future expansion of the treatment-eligible population. This figure, however, dramatically underestimates the total addressable market for a safe, effective, subcutaneous disease-modifying therapy, particularly one that could be used in combination with anti-amyloid agents.
Consider the comparison drug class: GLP-1 receptor agonists. Prior to 2020, GLP-1 agonists were a multi-billion-dollar diabetes class. Once the weight loss indication was established and subcutaneous delivery was optimized, they became the fastest-growing drug category in pharmaceutical history, approaching $100 billion in global annual revenue by the mid-2030s. The entire GLP-1 revolution was predicated on subcutaneous delivery making a pharmacologically effective therapy patient-accessible.
Alzheimer’s disease has 6.7 million American patients today and 13 million projected by 2050. If an effective anti-tau therapy were available subcutaneously, available to patients at the MCI stage, and combinable with anti-amyloid therapy, the market penetration could dwarf current anti-amyloid drug uptake. Even capturing 20% of the AD patient population at a WAC of $20,000-$30,000 annually would generate $40-80 billion in U.S. revenue alone.
This is not a base case projection. It is a ceiling analysis to illustrate the scale of what is at stake. The point is not that ARO-MAPT will capture this entire market. The point is that the opportunity is so large that even a modest share would generate revenues that justify a valuation far above Arrowhead’s current $8 billion market capitalization.
The Full Tauopathy Market and Arrowhead’s Indication Ladder Strategy
Beyond Alzheimer’s, the tauopathy spectrum includes PSP, CBD, FTD, CTE, and Pick’s disease, diseases that are devastating, entirely unaddressed by any disease-modifying therapy, and share the same molecular driver that ARO-MAPT is designed to silence. But the more important insight is not that these diseases represent additional market opportunity. It is that Arrowhead’s established regulatory strategy suggests they will pursue these smaller indications first, and use them as the clinical and regulatory launchpad for Alzheimer’s.
This is not speculative. It is the explicit operational blueprint Arrowhead has executed across every major program in its pipeline. The playbook has a name: the indication ladder. Identify the most severe, most clinically homogeneous, most genetically defined subpopulation of a disease, pursue orphan designation and accelerated approval there first, then climb to the larger indication with a validated drug, a clear regulatory pathway, and years of real-world safety data already accumulated.
The Plozasiran Template
The cleanest illustration of this strategy is plozasiran itself. Arrowhead did not wait for FCS approval before initiating sHTG trials; both programs ran in parallel. But they did not launch them at the same time with equal priority. They launched first into familial chylomicronemia syndrome (FCS), an ultra-rare genetic disorder affecting an estimated 3,000 to 5,000 patients in the United States. FCS patients carry loss-of-function mutations in lipoprotein lipase (LPL) and have triglycerides so elevated that no other standard lipid-lowering drug class works. They are pharmacologically abandoned by conventional therapies. In that population, plozasiran’s mechanism of silencing APOC3, which reduces triglycerides independently of LPL function, is not just effective; it is transformative. Ionis’s Tryngolza (olezarsen) is approved in FCS and is advancing toward sHTG approval as well, making it a real competitor in both indications. But the FCS market offered structurally superior trial economics regardless: a smaller, more homogeneous patient population requiring fewer subjects to power a trial, extreme disease severity producing large and measurable treatment effects, Orphan Drug Designation, Priority Review, and an accelerated regulatory pathway. Plozasiran earned its FCS approval on the strength of superior efficacy data, with the PALISADE Phase 3 trial (published in NEJM, Watts et al. 2024) showing roughly 70-80% triglyceride reduction versus approximately 59% for olezarsen in comparable trials, along with the quarterly dosing advantage of an RNAi therapeutic over a monthly ASO. The November 2025 FDA approval of Redemplo for FCS, secured faster than the larger sHTG program could have been, became the regulatory and commercial foundation for everything that followed.
The FCS trial completed and achieved approval well ahead of the sHTG program, not because sHTG was deprioritized, but because the FCS trial was structurally faster. The patient population was smaller and more homogeneous, the biomarker endpoints were cleaner and required fewer patients to achieve statistical significance, disease severity was more extreme and therefore detectable changes were larger, and the FDA’s orphan review pathway moved faster. By the time the sHTG program needed its most critical regulatory interactions, Arrowhead had a commercial product on the market, a real-world safety database, an established FDA relationship for the modality, and Breakthrough Therapy Designation for sHTG itself, in part because the agency had already seen plozasiran’s mechanism at work in the related population. The trials ran in parallel. The smaller indication just crossed the finish line first.
But the plozasiran story does not end at FCS and sHTG. Arrowhead’s ambition for this single molecule, silencing one gene, APOC3, extends across a full spectrum of triglyceride-related cardiovascular disease. Per the company’s Breakthrough Therapy Designation announcement (December 2025), the SUMMIT program currently encompasses four Phase 3 trials targeting progressively larger patient populations: SHASTA-3 (NCT06347003) and SHASTA-4 (NCT06347016) for sHTG (TG ≥500 mg/dL), MUIR-3 (NCT06347133) for mixed hyperlipidemia (TG 150-500 mg/dL, approximately 1,450 patients, targeting those with elevated ASCVD risk), and the CAPITAN cardiovascular outcomes trial. The durability of the plozasiran mechanism is supported by two-year open-label extension data presented at ACC.26 (March 2026), showing 83% median TG reduction in sHTG patients with no adjudicated acute pancreatitis events in any patient on plozasiran over the two-year period. CAPITAN is the plozasiran equivalent of a CVOT; if successful, it would position Redemplo not merely as a triglyceride-lowering drug but as a cardiovascular risk-reduction therapy in a population numbered in the tens of millions.
The progression from FCS to sHTG to mixed hyperlipidemia to a cardiovascular outcomes trial is the complete expression of the indication ladder strategy. One molecule. One gene. One delivery mechanism. Four indications, each larger than the last, each built on the regulatory and commercial infrastructure of the one before it. The patient population expands from 3,000-5,000 (FCS) to 2+ million (sHTG) to tens of millions (mixed hyperlipidemia) to the full addressable ASCVD-risk population, which could eventually encompass more than 100 million Americans alone. This is not gradual market expansion. It is a systematic, pre-planned climb from the most medically severe and commercially concentrated population to the broadest addressable market in cardiovascular medicine.
The ARO-MAPT Parallel: The Same Strategy, the Largest Prize
The plozasiran indication ladder maps almost perfectly onto what the ARO-MAPT indication ladder could become, and the parallel deserves to be made explicit. Where plozasiran started with FCS and is climbing toward cardiovascular outcomes, ARO-MAPT would start with PSP/CBD and is aimed ultimately at Alzheimer’s, the largest and most commercially consequential neurodegenerative disease market in existence. The structural template is identical: rare, severe, orphan-eligible disease first; well-characterized biomarker endpoints; accelerated regulatory pathway; then build outward to larger and larger populations using the safety database, commercial infrastructure, and established FDA relationships from earlier approvals.
There is one dimension in which the ARO-MAPT ladder is potentially even more powerful than plozasiran’s: the size of the ultimate destination. The CAPITAN cardiovascular outcomes trial targets patients with mixed hyperlipidemia and ASCVD risk, a large and commercially important population, but one already served by statins, PCSK9 inhibitors, and other lipid therapies. The Alzheimer’s Phase 3 destination for ARO-MAPT targets a disease where the current standard of care, anti-amyloid therapies with modest efficacy and significant side effects, leaves most patients with profound unmet need. An effective, subcutaneous, quarterly-dosed tau-silencing therapy that can be combined with anti-amyloid agents would not be competing for market share in an established drug class. It would be creating a new standard of care in a disease that has resisted it for more than a century. Plozasiran is the proof of concept. ARO-MAPT is the larger version of the same bet.
Applying the Ladder to Tauopathies: PSP and CBD First
Now apply that same template to ARO-MAPT. The analogous FCS-equivalent indications in the tauopathy space are Progressive Supranuclear Palsy (PSP) and Corticobasal Degeneration (CBD). These are two of the most devastating and neglected diseases in neurology. PSP affects approximately 20,000 to 30,000 patients in the United States; CBD affects fewer still. Both are invariably fatal, typically within 7-10 years of symptom onset. Both are dominated by 4-repeat tau pathology, the same tau species that ARO-MAPT targets. Both lack any approved disease-modifying therapy. Both qualify for Orphan Drug Designation, which confers seven years of market exclusivity, priority review eligibility, FDA fee waivers, and accelerated clinical pathway access.
It is worth pausing on the human weight of what that means. PSP is not an abstract disease category to this author. A childhood friend, someone whose company, humor, and full life were known to me personally, was recently taken by PSP. The disease progressed with the brutal efficiency it is known for: a year or two of subtle warning signs, then the rapid loss of balance, speech, and gaze control, then death. There is no approved disease-modifying therapy. There has never been one. Every family that has watched PSP take someone they love has faced a clinical reality unchanged since the disease was first described in 1964. That is not a footnote in this analysis. It is the entire reason the analysis matters. ARO-MAPT is not just an investment opportunity. It is, potentially, a way to end that story for the next family in line.
The critical structural advantage PSP and CBD offer is not just regulatory incentive; it is trial duration. PSP is a rapidly progressive disease. Median survival from symptom onset is 5-7 years, and functional decline is measurable within 12-18 months of a well-designed trial. This means a PSP Phase 2/3 trial can achieve statistically meaningful endpoints in 18-24 months of active treatment, compared to 36-48+ months for an Alzheimer’s Phase 3 trial where progression is slower and endpoints require longer observation. Faster progression equals faster trials. Faster trials equal faster approval, and because these trials run in parallel with the Alzheimer’s program, not instead of it, the net effect is that Arrowhead could hold an approved ARO-MAPT product in PSP while its Alzheimer’s Phase 3 is still enrolling. This is the exact dynamic that played out with plozasiran: FCS and sHTG ran simultaneously. FCS finished first. The commercial and regulatory benefits of that first approval then accelerated everything that followed.
PSP and CBD are also pharmacologically ideal for proving ARO-MAPT’s mechanism. Because they are pure or near-pure 4-repeat tauopathies, a clinical trial in PSP would produce a much cleaner signal-to-noise ratio for tau-related biomarker changes than a trial in Alzheimer’s, where multiple overlapping pathologies (amyloid, tau, neuroinflammation, synaptic degeneration) complicate interpretation. PSP has well-validated biomarkers, including plasma NfL, CSF total tau, CSF p-tau217, and MRI measures of superior cerebellar peduncle atrophy. The FDA has already engaged with PSP drug development in the context of prior ASO and antibody programs, meaning the regulatory framework is more established than it would be in a de novo rare disease.
PSP is to ARO-MAPT what FCS was to plozasiran: a pure, pharmacologically homogeneous disease with rapid progression, clean biomarker endpoints, small patient population, and full orphan regulatory incentives, all of which translate to a trial that runs faster, costs less, and crosses the finish line while the Alzheimer’s program is still in progress.
Why BIIB080 Has a Structural Ceiling for PSP and CBD
The deep brain distribution analysis presented in the competitive landscape section of this paper has a direct and decisive implication for the PSP and CBD indication ladder that is worth stating plainly: BIIB080 is not a credible competitor in these indications, and ARO-MAPT is. This is not a commercial or pricing argument. It is a neuropathology argument grounded in anatomy.
PSP is a disease of the subthalamic nucleus, the substantia nigra pars reticulata, the globus pallidus, the superior colliculus, the periaqueductal gray matter, and the rostral interstitial nucleus of the medial longitudinal fasciculus (riMLF). The riMLF is the structure whose tau-driven degeneration produces PSP’s most distinctive clinical sign: loss of voluntary vertical gaze. These are all deep, periventricular or parenchymal structures that sit at the far end of the intrathecal delivery gradient. The brainstem nuclei and subthalamic nucleus receive the lowest drug concentrations from a lumbar injection. BIIB080’s NHP knockdown data covers frontal cortex and hippocampus. It does not cover any of the structures that matter most in PSP.
CBD adds astrocytic plaques and coiled bodies in the cortex and basal ganglia to the lesion profile, but its defining subcortical degeneration also involves the substantia nigra, subthalamic nucleus, and putamen. The putamen in ARO-MAPT’s NHP data received 0.68 μg/g trigger concentration, within 2.3x of the most highly concentrated region. The substantia nigra received 0.47 μg/g. Both produced 70–80% MAPT mRNA knockdown in the same dataset. These are not theoretical claims. They are measured results in cynomolgus macaques.
The clinical consequence is this: a Phase 2 PSP trial with BIIB080 would be testing whether a drug that is well-characterized in cortex and hippocampus but under-characterized in subthalamic nucleus and brainstem can produce measurable clinical benefit in a disease defined by subthalamic nucleus and brainstem degeneration. The CSF tau biomarker may look good. The PSP Rating Scale endpoints, gaze palsy progression, postural instability, and dysphagia, may not respond, because the structures driving those symptoms are not the ones being reliably drugged. Biogen has not announced a PSP program for BIIB080. That silence may reflect exactly this constraint.
ARO-MAPT does not have this constraint. The 70–80% MAPT mRNA knockdown documented in the pons, the medulla, the thalamus, and the structures surrounding the substantia nigra in the CNS Delivery Summit dataset addresses the disease where the disease actually is. This is not an incremental advantage. For PSP and CBD, it is the difference between a program that could work and one that is pharmacologically equipped to.
BIIB080’s intrathecal delivery limits it to indications where the primary pathology is cortical and hippocampal. ARO-MAPT’s vascular distribution gives it access to every tauopathy, regardless of where in the brain the pathology is concentrated. PSP and CBD, the orphan-designated, rapidly progressing, first-rung indications on the tauopathy ladder, are precisely the indications where BIIB080’s delivery physics work against it and ARO-MAPT’s work in its favor.
Frontotemporal dementia caused by MAPT mutations (FTD-MAPT) represents an even more compelling early-stage target. Approximately 10-20% of familial FTD cases are caused by dominant mutations in the MAPT gene itself, the exact gene ARO-MAPT is designed to silence. These patients and their at-risk family members can be identified years or even decades before symptom onset through genetic testing. MAPT mutation carriers will develop disease. It is not a question of if; it is a question of when.
This creates a clinical trial opportunity that is, in many ways, even more powerful than the PSP opportunity: a prevention trial. If ARO-MAPT can be administered to presymptomatic MAPT mutation carriers and delay or prevent the onset of FTD, the regulatory and commercial implications are enormous. The FDA has been increasingly receptive to presymptomatic trials in genetic diseases, particularly since the DIAN-TU trials established the framework for intervening in familial Alzheimer’s before symptom onset. An FTD-MAPT prevention trial would be scientifically cleaner than any Alzheimer’s trial ever run: you know exactly who will get the disease, exactly what protein is driving it, and you have a targeted molecular intervention against that protein’s gene. The trial design writes itself.
Pricing in this context would be analogous to other precision medicine genetic disease therapies: Spinraza in SMA, Trikafta in CF, patisiran in hereditary ATTR amyloidosis. Those are $100,000 to $300,000 per year therapies. FTD-MAPT drug pricing at those levels, for a smaller patient population, generates meaningful revenues while establishing the clinical template for the far larger Alzheimer’s indication that follows.
CTE: The Emerging Population and the Sports Medicine Angle
Chronic traumatic encephalopathy (CTE) represents a longer-horizon but potentially enormous opportunity within the tauopathy ladder. CTE is caused by repetitive head trauma and is characterized by perivascular tau deposits in specific brain regions. It is estimated to affect millions of current and former contact sport athletes (NFL, NHL, boxing, rugby, soccer), military veterans, and others with histories of repeated head impacts. There is currently no approved therapy, no validated biomarker for antemortem diagnosis, and no disease-modifying treatment. But biomarker science is advancing rapidly: plasma and CSF p-tau217 and p-tau231 are being studied as potential CTE biomarkers, and PET imaging with tau tracers is increasingly used in research settings.
ARO-MAPT, if effective in established tauopathies, would be a logical candidate for CTE prevention or treatment trials. The addressable population, athletes, veterans, and others with documented head trauma history who are now at elevated tau accumulation risk, could ultimately number in the millions. A CTE indication would be a commercial opportunity unlike anything in the current neurology drug landscape, with strong advocacy tailwinds from professional sports leagues, veterans organizations, and the military.
The Parallel Execution Model: All Trials Running Simultaneously
The word ‘ladder’ is sometimes misread as implying sequence: finish one rung before stepping to the next. That is not how Arrowhead operates and it is not how this strategy works. The correct mental model is parallel pipelines with staggered finish lines. Once Phase 1 dose-optimization data confirm delivery, safety, and an appropriate dose range, Arrowhead would initiate trials across multiple tauopathy indications simultaneously. The PSP/CBD trials and the Alzheimer’s Phase 3 all run at the same time. What differs is when each one finishes, and those differences are structural, not strategic:
PSP/CBD Phase 2/3: Smaller patient populations (faster enrollment), rapid disease progression (endpoints reached in 18-24 months), orphan regulatory pathway (Priority Review shortens FDA clock by ~4 months), biomarker-based accelerated approval available. Expected approval timeline: 3-4 years post-trial initiation.
FTD-MAPT Prevention Trial: Genetically defined presymptomatic patients, genetic biomarkers as surrogate endpoints, FDA receptive to presymptomatic design precedents. Expected approval timeline: 4-5 years post-trial initiation.
Alzheimer’s Phase 2b then Phase 3: The most probable path requires a dedicated Phase 2b (approximately 150–300 patients, 12–18 months) to confirm dose selection and biomarker-endpoint alignment before FDA will sanction a 1,500–2,000 patient Phase 3. An adaptive seamless Phase 2b/3 design, increasingly common in AD and receptively received by FDA under Breakthrough Therapy Designation, can eliminate the gap between phases. Expected approval timeline: 8–10 years post-Phase 1 data on the standard path; 6–8 years if an adaptive seamless design is accepted. Crucially, this timeline runs in parallel with PSP and CBD, not in sequence.
The timeline differential means that by the time the Alzheimer’s Phase 3 is at the midpoint of its blinded treatment period, Arrowhead may already hold an FDA-approved ARO-MAPT product generating commercial revenue in PSP and CBD. That commercial infrastructure, the prescriber network, the reimbursement contracts, the real-world safety database, the manufacturing scale, does not sit idle. It gets deployed for Alzheimer’s the moment the Phase 3 reads out, and the Alzheimer’s Phase 3 itself benefits directly from the PSP data: the FDA has already reviewed ARO-MAPT’s safety and mechanism of action in a tau disease, which shortens the review clock and increases the probability of approval.
This is the strategy in full. The smaller indications are not stepping stones that must be cleared before Alzheimer’s can begin. They are parallel programs running on faster tracks, faster because the diseases progress faster, the patient populations are smaller, and the regulatory incentives are stronger. The Alzheimer’s program does not wait for them. But it benefits enormously when they arrive first.
Arrowhead does not need to win Alzheimer’s to create extraordinary near-term value. PSP and CBD trials running in parallel with the AD program means potential orphan approvals, with premium pricing, zero competition, and an established regulatory track, could generate commercial revenue and platform validation years before the Alzheimer’s readout. The smaller indications cross the finish line first. The largest prize is still in the race.
IX. Investment Thesis: What Positive 2H 2026 Data Means for Arrowhead
Where Arrowhead Stands Today
Arrowhead enters the ARO-MAPT data window from a position of significant strength. Per its fiscal Q1 2026 results (ended December 31, 2025), the company had triggered a $200 million Sarepta milestone (ARO-DM1 enrollment target), closed the $200 million Novartis upfront for ARO-SNCA, received Breakthrough Therapy Designation for plozasiran in sHTG, initiated the ARO-MAPT Phase 1/2a, and reported approximately $1.09 billion in trailing twelve-month revenues. The company has demonstrably crossed the threshold from binary-risk biotech to multi-product RNA therapeutics platform with significant and growing cash generation.
Arrowhead’s balance sheet at the Q1 2026 reporting date was structurally sound for the catalyst-rich period ahead. Per the Q1 2026 earnings report, the company reported cash and equivalents sufficient to fund operations through the 2H 2026 data window and well beyond, supported by ongoing Redemplo FCS commercial revenue, the Novartis collaboration payments, and anticipated plozasiran sHTG milestones. The $200 million Novartis upfront closed in October 2025 and the $200 million Sarepta milestone triggered in November 2025, together adding $400 million in non-dilutive cash in the six months prior to the ARO-MAPT data readout. Dilution risk is not a material concern for investors modeling the 2H 2026 to 2028 window. Arrowhead does not need to raise capital to reach its most important near-term milestones, and the pipeline itself is generating the cash to fund them.
The stock traded at a 52-week low of $9.57 before the series of catalysts in 2025 drove it to a high approaching $77. It currently trades near $60-65 with a market capitalization of approximately $8 billion and a consensus analyst rating of Strong Buy with a median price target near $82. Piper Sandler has a $110 price target.
The market is not ignoring Arrowhead. But neither is the market pricing ARO-MAPT’s optionality. The stock is being valued primarily on plozasiran (Redemplo) revenues, the Novartis collaboration, zodasiran, and the near-term cardiometabolic pipeline. ARO-MAPT’s contribution to the valuation is close to zero in the base case today. A positive Phase 1/2a readout would add value from a standing start of near-zero market pricing.
The Three Layers of Value Creation
Layer 1: ARO-MAPT as a Drug Asset
Positive Phase 1/2a data demonstrating human tau knockdown with subcutaneous ARO-MAPT would immediately establish it as the lead clinical candidate in the tau space on delivery modality alone. The nearest analog for valuation purposes is BIIB080 itself: Biogen paid Ionis $1.5 billion upfront at a time when BIIB080 had Phase 1/2 data showing intrathecal tau reduction of 56–60% in human CSF. ARO-MAPT enters Phase 1 with NHP data showing 70–80% MAPT mRNA knockdown across all brain regions and 50–60% CSF tau protein reduction via subcutaneous injection, first presented publicly at the 7th Annual CNS Delivery Summit on December 10, 2025. If human data reflects the NHP results, ARO-MAPT would achieve comparable tau knockdown to BIIB080 via a fundamentally superior delivery route. A drug that does what BIIB080 does, but subcutaneously, with deep brain distribution, and quarterly dosing, would be worth meaningfully more than the $1.5 billion Biogen paid for the intrathecal version. Applied to Arrowhead’s current market cap, a licensing transaction in that range alone would represent a 15-25% uplift on the drug asset, before any platform premium is applied, and that underestimates the competitive bidding dynamic. Biogen knows better than anyone what validated tau knockdown data is worth. So does Eli Lilly, which already has an intrathecal tau siRNA (LY-3954068) in Phase 1 and would have every incentive to access a subcutaneous solution to complete its tau strategy. If ARO-MAPT’s Phase 1/2a data shows subcutaneous tau reduction matching the NHP benchmark, Lilly would likely be among the first in the room.
Layer 2: The CNS Delivery Platform
This is where the real value lives, and it is where the market is most underpricing Arrowhead today. ARWR is not a single-asset company, and ARO-MAPT is not a standalone program. It is the clinical validation event for a CNS delivery system that can be applied to every neurological target the TRiM platform can address. After positive ARO-MAPT data, the pipeline that becomes immediately activatable includes: the Novartis ARO-SNCA milestones (already licensed, but human delivery validation unlocks milestone payments), ARO-HTT (SRP-1005, licensed to Sarepta, CTA filed Q1 2026) and ARO-ATXN2 (also licensed to Sarepta, targeting ataxin-2 for SCA2 and ALS-TDP-43) are already in Arrowhead’s partnered CNS pipeline and generate milestones as they advance; the Novartis ARO-SNCA milestones unlock further as human delivery is validated; and potential future internal or partnered programs on targets such as SOD1 and TDP-43 in ALS become immediately credible once the platform is proven in humans. Each validated CNS target is not a new drug development bet; it is an incremental expression of a delivery technology that will have already been proven in humans. The hard problem gets solved once. The programs multiply from there.
The value of a validated, proprietary subcutaneous CNS delivery platform in the current drug development environment is not measured in hundreds of millions. It is measured in tens of billions. Alnylam built a $40 billion market capitalization primarily on the back of its GalNAc-siRNA liver delivery system. The CNS delivery problem is categorically harder, the market for CNS diseases is larger by an order of magnitude, and the competitive field of companies with validated subcutaneous CNS RNAi delivery is, at the moment of this writing, exactly zero. If ARO-MAPT changes that number to one, Arrowhead owns one of the most valuable proprietary delivery platforms in the history of drug development, and every company currently developing CNS gene silencing via intrathecal delivery, including Alnylam with its entire five-program CNS pipeline, faces a structural re-evaluation. The platform value of validated subcutaneous CNS RNAi is not contained within Arrowhead’s own programs. It radiates outward to reshape the decision calculus of every large pharma company that has, or wants to have, a CNS gene silencing strategy, and every company currently developing a CNS gene silencing program via intrathecal delivery, including Alnylam with its entire five-program CNS pipeline, faces a structural re-evaluation of their delivery strategy. The platform value of validated subcutaneous CNS RNAi is not contained within Arrowhead’s own programs. It radiates outward to reshape the decision calculus of every large pharma company that has, or wants to have, a CNS gene silencing strategy.
Layer 3: Acquisition Premium
Arrowhead is an acquisition target. It has been an acquisition target since the TRiM platform was first validated in the liver. With Redemplo approved and generating revenue, a clinical-stage CNS program, a Novartis collaboration, and multiple late-stage assets in cardiology and metabolic disease, the strategic value to a large-cap pharmaceutical acquirer is substantial and growing.
Positive ARO-MAPT data would likely accelerate acquisition interest from the major players already active in Alzheimer’s drug development: Eli Lilly (the current market leader in Alzheimer’s with Kisunla and an enormous AD pipeline), Biogen (deeply invested in tau via BIIB080 and acutely aware of the subcutaneous delivery advantage), Roche/Genentech (the most diversified Alzheimer’s portfolio), and Pfizer, AstraZeneca, or others looking for a fast path into neurology via platform acquisition. In a takeout scenario, the premium to current market cap could be transformational, potentially 50-150% or more depending on the quality of the data and the competitive bidding environment, consistent with prior CNS platform acquisitions where validated delivery technology commanded multiples of pre-announcement market value.
X. The Analyst Blind Spot: Why the 13 Official Analysts Are Missing the Biggest Variable
The Official Coverage Universe
Arrowhead Pharmaceuticals lists 13 sell-side firms on its official investor relations analyst coverage page. These are the analysts whose research shapes the institutional investment community’s view of the stock. They are talented, diligent, and deeply familiar with Arrowhead’s pipeline. They are also, collectively, working with a valuation framework that is structurally incapable of pricing the most important thing Arrowhead may do in 2026. Understanding the specific shape of that blind spot, analyst by analyst, thesis by thesis, is essential context for the ARO-MAPT data thesis.
Reading the Spread: What $35 to $110 Actually Tells You
The Q1 2026 earnings cycle illustrated this dynamic clearly. BofA’s Jason Gerberry trimmed his target from $84 to $81 after reducing the probability of success for obesity programs, while keeping a Buy rating anchored to the sHTG data. Leerink’s Mani Foroohar reiterated Market Perform at $61 following Sarepta’s data, focused entirely on near-term cardiometabolic catalysts. Meanwhile, TD Cowen reiterated Buy looking ahead to additional details in 2H 2026 including sHTG data. In every case, the analyst commentary centered on the cardiometabolic and obesity pipeline. ARO-MAPT and the CNS platform were not the focus. They rarely are. That is the gap.
Bernstein’s $35 target is particularly telling. With ARWR trading at $60-65, Bernstein is arguing that the stock is nearly 50% overvalued, not because the programs don’t work, but because they don’t assign value to what the programs collectively represent. The same pattern is visible at Leerink ($61) and Morgan Stanley ($78). These are not negative views on Arrowhead’s science. They are views anchored to rNPV methodology applied to a company whose primary value is now increasingly being set by strategic acquirers and large-pharma partners who price delivery platforms, not drug pipelines.
Consider the Novartis deal again from this lens. Novartis paid $200 million upfront for preclinical ARO-SNCA rights: rights to one program, on one target, using one tissue-type delivery system, with no human data. At the time of that deal, the CNS delivery platform had NHP data but nothing in humans. Since then, Arrowhead has disclosed that ARO-MAPT, using the same delivery technology, achieved 70–80% MAPT mRNA knockdown across all NHP brain regions (up to 85% in cortex) AND 50–60% NHP CSF tau protein reduction maintained for 5 months, matching the CSF tau reduction range that BIIB080 achieved in human Phase 1b patients via lumbar puncture. That figure matches the intrathecal benchmark set by BIIB080, the most advanced tau-targeting program in the field, but was achieved without a lumbar puncture. A standard rNPV model for ARO-MAPT at Phase 1 still assigns this minimal value. Novartis has already told the market what this platform is worth at preclinical stage. The NHP figure now disclosed makes the argument even more concrete. The fact that a $35 price target persists in this environment is the clearest illustration of the valuation gap this section is identifying.
What Is Absent From Every Model on the Street
Across all 13 official coverage firms, the following value drivers are either absent entirely from financial models or assigned de minimis rNPV. This is not speculation, it is observable from the fact that no analyst commentary on ARO-MAPT references platform-level value creation or parallel tauopathy indication strategies:
The SRP-1005 and ARO-ATXN2 Signal That None of the 13 Are Talking About
Buried in the Q1 fiscal 2026 earnings call transcript is one of the most underappreciated disclosures in Arrowhead’s recent history. CEO Christopher Anzalone confirmed that Sarepta Therapeutics has submitted a Clinical Trial Application for SRP-1005 (formerly ARO-HTT) for the treatment of Huntington’s disease, using the same subcutaneous blood-brain barrier delivery system as ARO-MAPT. What that disclosure did not amplify is that Sarepta has also licensed ARO-ATXN2, a second CNS program targeting ataxin-2 for spinocerebellar ataxia type 2 and ALS, on the same delivery platform. This means there are now three clinical-stage or advanced-preclinical CNS programs on the TRiM BBB platform from two separate large-pharma partners: ARO-MAPT (Arrowhead, wholly owned), SRP-1005 (Sarepta, CTA filed Q1 2026), and ARO-ATXN2 (Sarepta, licensed). None of the 13 analysts covering Arrowhead have published material commentary assigning standalone platform-validation value to either Sarepta CNS program.
Sarepta did not license SRP-1005 and ARO-ATXN2 because of a relationship favor. They licensed two separate CNS programs because their scientific diligence concluded the TRiM BBB delivery technology was credible enough to commit to two of the most genetically airtight targets in neurology. SRP-1005 targets HTT: every person who carries the Huntington’s mutation will develop the disease. ARO-ATXN2 targets ataxin-2, a genetic modifier of TDP-43 toxicity that dramatically amplifies ALS and SCA2 risk in carriers of ATXN2 repeat expansions. An siRNA that silences HTT or ATXN2 throughout the brain from a subcutaneous injection would be transformative for patients in each indication and commercially extraordinary. Two CTA-equivalent filings from the same partner are two declared votes of confidence in the platform. Neither is in the models.
The 13 official analysts have price targets ranging from $35 to $110, a $75 spread on the same stock. The divergence is not about the cardiometabolic pipeline, which most analysts understand well. It is entirely about whether you assign value to the CNS delivery platform or treat it as a speculative phase 1 footnote. Novartis paid $200M upfront to answer that question for a single preclinical CNS program. Sarepta answered it with a CTA submission. The 13 analysts haven’t answered it yet. The 2H 2026 ARO-MAPT data will force them to.
The Short Interest as a Contrarian Indicator
As of early 2026, short interest in ARWR stands at approximately 11.2 million shares, roughly 8.3% of the float, with 5.7 days to cover. For a stock with a commercial-stage product, $1B+ in trailing revenues, Strong Buy consensus from the majority of its 13 official analysts, and a binary CNS data catalyst in 2H 2026, that level of short interest is elevated. It represents institutional capital betting that the platform thesis will not be validated before the current valuation becomes untenable.
The short argument is not that Arrowhead is a fraud or that the science is wrong. The short argument is that rNPV-based fair value is below the current stock price, and that the 2H 2026 data will disappoint or be insufficient to force model revisions. That is a coherent position. It is also the same position that was coherent at $30, $40, $50, and $60 as the stock climbed through each level. Short sellers who have maintained that framework have been repeatedly stopped out by newsflow that a traditional rNPV model doesn’t price in advance. The 2H 2026 readout is the next such event.
The Re-Rating Has Not Happened Yet. Here Is Why It Will.
Platform re-ratings in biotech are discontinuous, not gradual. Alnylam did not gradually drift from pipeline company to platform company as analysts slowly raised models. It re-rated sharply when patisiran’s Phase 3 data validated GalNAc-siRNA liver delivery as a reproducible technology. Sarepta re-rated when exon 51 skipping data validated the DMD franchise. Each time, the street’s models changed not because analysts evolved their thinking but because a single event made the old framework untenable. ARWR’s re-rating from a cardiometabolic RNA company to a multi-tissue CNS platform company has not happened. The 2H 2026 data readout is the forcing function.
If the data shows subcutaneous tau knockdown in humans consistent with the 70–80% NHP mRNA and 50–60% NHP CSF tau benchmarks, the number of analysts who can justify a $35 price target drops toward zero. The number who can explain why a program that has just demonstrated in humans what BIIB080 demonstrated via lumbar puncture, but administered subcutaneously with uniform deep brain distribution, is worth less than the $1.5 billion Biogen paid for the intrathecal version also drops toward zero. The five or six analysts with targets in the $35-$78 range, anchored to rNPV methodology that assigns a Phase 1 CNS program $1-3 per share, will be forced to rebuild models against a program that is now clinically validated. Even David Bautz at Zacks SCR, who has been one of the most systematic and granular ARWR fundamental analysts and whose most recent report already acknowledges the CNS platform thesis, has a $78 target that does not yet price in human delivery validation. Price targets will move. Short covering will amplify the move. The institutional investors who understand platform inflections and positioned ahead of the data will be well compensated for the patience. That the 13 official analysts have not yet converged on this thesis is not a warning sign. It is the opportunity.
XI. Scenario Analysis: Bull, Base, and Bear Cases for 2H 2026 Data
The following scenarios represent a framework for thinking about the range of outcomes from the 2H 2026 initial data readout. They are not price targets or investment recommendations. They are analytical constructs for thinking through probability-weighted outcomes. The key reference points for calibrating human data: ARO-MAPT achieved greater than 75% MAPT mRNA knockdown in NHP CNS tissue at clinically translatable doses; BIIB080’s comparable NHP intrathecal data of 74–77% translated to 56–60% CSF tau protein reduction in humans. A similar NHP-to-human translation for ARO-MAPT would imply approximately 55–60%+ CSF tau reduction. Any result in that range or above constitutes validation. Any result materially above 60% with evidence of deep brain distribution would be exceptional.
BULL CASE | The Subcutaneous Revolution Confirmed | Probability: 35%
ARO-MAPT achieves ≥65% reduction in CSF tau biomarkers in healthy volunteers, consistent with or exceeding the NHP-to-human translation ratio established by BIIB080
PK data confirms uniform CNS distribution after subcutaneous dosing, with evidence of deep brain structure penetration that intrathecal delivery cannot match
Early AD cohort data shows matching or superior tau knockdown with a clean safety profile
Plasma p-tau217 confirms blood-based biomarker response, opening the door to monitoring without lumbar puncture
Management confirms plans for a PSP or CBD orphan indication trial, activating the indication ladder
Dosing interval data is consistent with quarterly subcutaneous administration; patient experience advantage over BIIB080 is quantifiable
Novartis ARO-SNCA milestones triggered by human data; Biogen, Lilly, or another major pharma announces partnership or acquisition discussion
Analyst consensus target prices move to $120-$150+ range; the rNPV framework for a pipeline company becomes untenable
Stock reaction: +50% to +150% on data day; sustained multi-year rerating as indication ladder unfolds, platform partnerships accelerate, and acquisition premium expands
BASE CASE | Validation Without the Superlative | Probability: 45%
ARO-MAPT achieves 45-65% reduction in CSF tau biomarkers in healthy volunteers, directionally consistent with NHP-to-human translation
Safety profile is clean; no unexpected adverse events related to CNS delivery or off-target neurological effects
PK data confirms BBB penetration after subcutaneous dosing, establishing first-in-class human proof of concept
Management confirms progression to multiple-dose cohorts in AD patients; 2027 data window for deeper efficacy
Data is positive but early-stage; market waits for AD patient biomarker data before full platform re-rating
A partnership discussion is confirmed or advanced; deal terms comparable to or exceeding the Novartis ARO-SNCA deal
Analyst price targets rise to $90-$110 range on the basis of validated CNS delivery platform
Stock reaction: +20% to +40% on data day; gradual rerating as AD patient data matures and partnership discussions formalize
BEAR CASE | Delivery Doesn’t Translate | Probability: 20%
ARO-MAPT achieves <30% CSF tau reduction, well below the NHP-predicted range and below the therapeutically meaningful threshold
CNS drug concentrations are variable or insufficient in humans; the NHP BBB penetration does not translate
Safety signal emerges: neuroinflammation, off-target CNS effects, or delivery-related adverse events
No early AD patient data is ready; management signals that dose optimization is required before progression
Program placed on clinical hold or dose-escalation paused pending safety review
Market re-evaluates the entire CNS platform thesis; Novartis ARO-SNCA milestone cadence and the $200M upfront decision scrutinized
Stock reaction: -5% to -15% on data day; limited sustained downside given the strength of the non-CNS commercial pipeline, which independently supports the current valuation
The asymmetry here matters enormously. In the bear case, Arrowhead still has Redemplo generating commercial revenue, zodasiran in Phase 3, ARO-DIMER-PA, ARO-PNPLA3, the Novartis collaboration, and the Sarepta ARO-DM1 milestones. The downside is bounded by a highly productive non-CNS pipeline that independently justifies a multi-billion-dollar valuation. The upside in the bull case includes CNS platform validation, the entire tauopathy indication ladder from PSP to Alzheimer’s, and near-unlimited pipeline optionality across every CNS target the TRiM platform can address. This is not symmetric risk-reward. It is deliberately asymmetric, exactly the kind of setup long-term platform investors should seek.
A Data-Driven Prediction for ARO-MAPT Human Results
Predicting human clinical outcomes from preclinical data is inherently uncertain, and this section is offered as analytical context, not as a guarantee. However, the combination of ARO-MAPT’s NHP data and the established BIIB080 NHP-to-human translation framework provides a more rigorous basis for expectation-setting than is typical for a Phase 1 program entering its first data window. The following framework synthesizes both datasets into a grounded projection.
Step One: The BIIB080 Translation Template
BIIB080’s development trajectory established a reference point for how tau-targeting CNS programs translate from NHP to human. In cynomolgus macaques, intrathecal BIIB080 achieved 74% MAPT mRNA knockdown in hippocampus and 77% in frontal cortex. In the Phase 1b human trial (Mummery et al., 2023), the highest-dose groups achieved 56–60% CSF total-tau protein reduction. That represents a translation efficiency of approximately 74–79% from NHP brain tissue mRNA to human CSF protein: deep, durable knockdown in animals that translated reliably to meaningful protein reduction in patients.
Step Two: Applying the Framework to ARO-MAPT
ARO-MAPT’s NHP data introduces two additional inputs that BIIB080’s NHP package did not contain. First, ARO-MAPT achieved 70–80% MAPT mRNA knockdown across all brain regions (up to 85% in some cortex areas) via subcutaneous dosing, comparable in depth to BIIB080’s intrathecal NHP figures. Second, and more directly relevant, ARO-MAPT’s NHP CSF total-tau protein reduction was measured at 50–60%, maintained for up to 5 months. This second figure is the most important because it is the same biomarker that will be reported in human subjects: CSF total-tau percent reduction from baseline.
The key analytical insight is that BIIB080’s NHP-to-human translation step moved from brain mRNA reduction to human CSF protein reduction, a two-step translation involving species differences and biomarker conversion. ARO-MAPT’s NHP data has already completed that first step: the NHP CSF tau protein data is directly comparable to the clinical endpoint. The remaining translation question is simply whether NHP CSF tau reduction predicts human CSF tau reduction, and for that question the relevant precedent is the BIIB080 CSF-to-CSF comparison.
Step Three: The Projection
If the NHP-to-human CSF tau translation for ARO-MAPT follows a similar attenuation ratio to what BIIB080 demonstrated across species, a reasonable expectation would be a 15–25% step-down from the NHP CSF tau result of 50–60%. That produces a central estimate of approximately 38–51% CSF total-tau reduction in humans at the optimal dose. Two factors support a result at the higher end of this range or above it. The TfR1 mechanism is confirmed cross-reactive between cynomolgus macaques and humans, meaning the BBB-crossing ligand binds the same receptor in both species with similar affinity, and vascular delivery scales more predictably to the larger human brain than intrathecal delivery does: drug distributed via the bloodstream reaches each brain region in proportion to its vasculature, whereas intrathecal delivery depends on CSF flow dynamics that become less efficient in the larger human brain.
A further consideration is trial design. The 2H 2026 readout will likely report data from the early dose cohorts in healthy volunteers, where the full loading regimen of three weekly doses may not yet be complete in all subjects. Earlier cohorts receiving fewer doses will show more modest biomarker reductions. The most clinically relevant numbers, those from subjects who received the complete loading regimen at the higher doses, are the ones to benchmark against the NHP data.
Central projection: ARO-MAPT is expected to achieve approximately 40–55% CSF total-tau reduction in humans at the optimal loading dose in the 2H 2026 initial readout. A result of 40–55% would confirm NHP-to-human translation and establish ARO-MAPT as producing comparable tau knockdown to BIIB080 via subcutaneous injection. A result above 55% would exceed BIIB080’s Phase 1b best and constitute an exceptional outcome. A result below 30% would fall materially short of NHP predictions and represent a translation failure. This projection is grounded in three inputs: ARO-MAPT’s NHP CSF tau data (50–60%), the BIIB080 NHP-to-human translation ratio, and the TfR1 species cross-reactivity confirmation. It is not investment advice and carries the inherent uncertainty of all clinical predictions.
XII. Platform Expansion: What the CNS Frontier Unlocks
Beyond Tau: The CNS Target Pipeline
If ARO-MAPT validates subcutaneous CNS delivery in humans, the pipeline implications are staggering. Consider the list of CNS targets for which genetic or preclinical evidence supports RNAi as a potential therapeutic approach:
Arrowhead has already licensed one of these programs (SNCA) to Novartis. If ARO-MAPT validates the CNS delivery system, every other target on this list becomes a fundable internal or partnershipable program. The company does not need to develop all of them in-house. It needs only to demonstrate that the platform works, and then license, collaborate, or spin out programs as the strategic situation dictates.
The Tissue Type Expansion Arc
Arrowhead’s history is one of systematic tissue type expansion, and each expansion has followed a recognizable pattern: identify a biologically validated target in a new tissue, develop a TRiM conjugate engineered for that cell type, prove delivery and knockdown in animals, advance to Phase 1. Liver came first and produced the most mature commercial pipeline. Lung followed. Muscle and adipose tissue opened obesity and metabolic disease programs. Solid tumors, specifically clear cell renal cell carcinoma via ARO-HIF2, proved that the platform is not confined to metabolic organs and can find and silence genes inside tumor cells. CNS is now in the clinic, and Arrowhead has already publicly signaled what comes next: the eye.
The Ocular Frontier: Trabecular Meshwork and Glaucoma
Arrowhead has presented preclinical data at the 16th European Glaucoma Society Congress (June 2024) and at TIDES Europe 2024 (November 2024) on a TRiM platform engineered for delivery to the trabecular meshwork via intracameral administration, an injection directly into the anterior chamber of the eye. The target tissue is the trabecular meshwork, the primary drainage structure responsible for regulating aqueous humor outflow and intraocular pressure (IOP). Dysfunction in the trabecular meshwork is the central driver of primary open-angle glaucoma, the world’s leading cause of irreversible blindness, affecting an estimated 80 million people globally.
The preclinical data presented show that the TRiM ocular platform achieves dose-dependent gene knockdown in rat trabecular meshwork cells at Day 15 post-intracameral injection, with the TRiM-conjugated molecule outperforming non-conjugated siRNA at every dose level. Critically, at a 50 μg per eye dose, knockdown was durable: 60-70% gene silencing was maintained across measurement timepoints at Days 15, 29, 43, and 57 after a single injection. For a disease currently managed with daily eye drops that patients routinely fail to instill correctly, non-adherence is estimated at 30-80% in glaucoma, a single intracameral RNAi injection achieving over two months of durable IOP-relevant gene knockdown would represent a clinically transformative improvement in the treatment paradigm.
The glaucoma market context is compelling. Current first-line agents include prostaglandin analogs (latanoprost, bimatoprost), beta-blockers, and the newer Rho kinase inhibitor netarsudil, all requiring daily topical dosing and burdened by the adherence problem. There is no approved RNAi therapy for any ocular indication. A trabecular meshwork-targeted TRiM therapeutic could silence relevant genes, including MYOC (myocilin, mutations in which cause a subset of familial glaucoma) or RHOA (a Rho kinase pathway target), with quarterly or potentially semi-annual dosing, administered as an office procedure. The commercial model would resemble intravitreal anti-VEGF injections for wet AMD: a well-established clinical workflow that retinal specialists already perform millions of times annually.
The ocular program is still preclinical. But the fact that Arrowhead is presenting this data publicly at ophthalmology conferences means it is being positioned for advancement. If ARO-MAPT validates the CNS delivery system in 2H 2026, the momentum behind the entire platform accelerates, and the ocular program moves from scientific poster to prioritized development candidate on a faster timeline. Every tissue type Arrowhead adds to its validated delivery repertoire is another commercial platform, another partnership opportunity, and another reason the company’s valuation ceiling keeps rising.
The Cardiac Frontier: Cardiomyocyte Delivery and the Next Billion Patients
The CNS Delivery Summit slide deck contained one entry that received almost no attention and deserves considerably more: cardiomyocyte delivery is listed as a preclinical TRiM platform. No data was presented, no target was disclosed, and no IND timeline was mentioned. But the inclusion of cardiomyocyte alongside liver, lung, skeletal muscle, CNS, adipose, and ocular in Arrowhead’s published tissue type roster signals that the company has identified a cardiomyocyte-targeting receptor and demonstrated some level of siRNA uptake in cardiac muscle cells. That alone is a meaningful disclosure given how hard cardiomyocyte delivery has proven historically.
Cardiomyocytes are among the most difficult cells to drug systemically. They are post-mitotic, heavily protected by extracellular matrix, have limited endocytic activity compared to hepatocytes, and sit behind a cardiovascular filtration system that clears most macromolecules before accumulation. Gene therapy approaches using AAV vectors have struggled with immunogenicity, manufacturing complexity, and re-dosing limitations. If Arrowhead has engineered a TRiM conjugate that achieves receptor-mediated uptake in cardiomyocytes after subcutaneous dosing, the potential therapeutic targets make the liver and CNS pipelines look modest in comparison.
The most compelling target is phospholamban (PLN). PLN encodes a small protein that inhibits SERCA2a, the calcium pump responsible for cardiomyocyte relaxation and a central regulator of cardiac contractile function. Gain-of-function PLN mutations cause inherited dilated cardiomyopathy. Loss-of-function PLN mutations in humans are associated with enhanced cardiac function without adverse consequence. De-inhibiting SERCA2a by silencing PLN has been the therapeutic goal of cardiac gene therapy programs for two decades. The CUPID trials attempted to deliver SERCA2a directly via AAV and failed on potency and durability. Silencing PLN with RNAi to achieve the same net effect, without delivering a viral vector or a transgene, is mechanistically cleaner and avoids the immunogenicity and re-dosing barriers that have plagued AAV approaches. Heart failure affects 6.7 million Americans. Peak annual revenue for an effective, subcutaneous, quarterly-dosed PLN silencer would be measured in tens of billions.
Hypertrophic cardiomyopathy (HCM) is the second major opportunity. Dominant gain-of-function mutations in MYH7 and MYBPC3, the genes encoding the major sarcomere proteins, cause HCM in approximately 1 in 500 people globally, making it the most common inherited cardiac disease. Bristol-Myers Squibb paid $13.1 billion for MyoKardia specifically because sarcomere modulation has commercial validity: mavacamten (Camzyos) proved the concept. Allele-selective RNAi silencing of the dominant mutant sarcomere allele, delivered subcutaneously to cardiomyocytes, would be a disease-modifying approach targeting the causal mutation rather than its downstream hemodynamic consequences. The genetic validation is as strong as any target in cardiovascular medicine.
A third candidate is transthyretin amyloid cardiomyopathy (ATTR-CM). The approved ASO tafamidis and the RNAi agents patisiran and vutrisiran address ATTR-CM systemically by silencing hepatic TTR production. But TTR is also expressed locally in cardiomyocytes, and emerging evidence suggests that local cardiac TTR production may contribute to amyloid deposition in the myocardium. A cardiomyocyte-targeted siRNA silencing local TTR production, used in combination with or instead of hepatic silencing, could produce more complete amyloid suppression at the site of pathology. ATTR-CM is dramatically underdiagnosed, estimated at 300,000 to 500,000 patients in the US and growing as diagnostic awareness improves.
No cardiomyocyte data has been publicly disclosed. Arrowhead has not named the receptor being targeted, the lead compound, or the indication. Based on Arrowhead’s established pattern of presenting each new tissue type at scientific conferences before filing INDs, the first cardiomyocyte platform data would most likely appear at the American Heart Association, the American College of Cardiology, or TIDES, potentially in 2026 or 2027. The investor signal to watch is any Arrowhead presentation at a cardiology conference: that event would confirm the cardiac platform is being positioned for advancement in the same way that the glaucoma conference presentations confirmed the ocular program.
The cardiomyocyte platform is Arrowhead’s most undisclosed frontier, and potentially its most valuable. If TRiM-mediated subcutaneous siRNA delivery to cardiomyocytes is validated in animals and subsequently in humans, it would open PLN-targeted heart failure, MYH7/MYBPC3-targeted hypertrophic cardiomyopathy, and local TTR silencing in ATTR-CM as addressable indications. Combined, these three indications represent a patient population and commercial opportunity that rivals the entire tauopathy spectrum. The CNS Delivery Summit’s single-line disclosure of “Cardiomyocyte: Preclinical stage” may prove to be one of the most consequential disclosures in Arrowhead’s history.
Each new tissue type conquest was not just a drug program. It was a platform expansion event that expanded the total addressable biology and the company’s strategic value. CNS is the largest expansion in Arrowhead’s history to date, but ocular and cardiac are not far behind. The potential market, the number of druggable targets, and the partnership value of validated delivery into these tissues are all substantial. The pattern that positive ARO-MAPT data will trigger a wave of new program announcements, internal and partnered, CNS, ocular, and cardiac, mirrors what happened when liver delivery was validated years ago, but at an order of magnitude greater scale.
XIII. The Scientific and Manufacturing Edge
A Team That Has Done the Impossible, Repeatedly
The thesis of this paper rests on a technical claim: that Arrowhead has engineered a subcutaneous delivery system capable of crossing the blood-brain barrier and silencing target genes throughout the CNS after a systemic injection. That is an extraordinary claim. The skepticism is warranted. But before dismissing the preclinical data on grounds of prior field failures, it is worth examining the track record of the people making that claim, because their history is not one of incremental science. It is one of solving delivery problems that were widely considered unsolvable.
The RNAi field in its early years was defined by a single frustration: the science worked brilliantly in the test tube and fell apart in the body. Naked siRNA was degraded in the bloodstream before reaching its target. Early delivery vehicles, lipid nanoparticles, polymer conjugates, viral vectors, were effective but toxic, immunogenic, or too complex to manufacture consistently. For much of the 2000s and early 2010s, it appeared that RNAi’s therapeutic potential might remain perpetually out of reach. Arrowhead’s scientific team did not accept that. They designed their way around it.
The TRiM platform that exists today is not the product of one clever insight. It is the product of more than a decade of iterative discovery chemistry: systematically identifying the minimum molecular architecture required to achieve receptor-mediated cellular uptake in a target tissue, then stripping away every unnecessary element to reduce toxicity, cost, and complexity. CEO Christopher Anzalone has described the TRiM design philosophy as finding ways to progressively trim away extraneous features and retain only what drives pharmacologic activity. That philosophy, applied relentlessly by the chemistry and biology teams, is what produced a platform capable of reaching cell types that no RNA therapeutic had previously targeted.
The Multi-Tissue Achievement: A Scorecard
The breadth of tissue-type delivery that Arrowhead has achieved is without parallel in the RNAi field. Each tissue type required solving a distinct set of delivery, uptake, and pharmacology challenges, challenges so significant that most companies focus on one tissue for years. Arrowhead has validated clinical delivery in multiple tissues simultaneously, and the potency of knockdown at each site is not marginal. It is routinely among the deepest gene silencing ever documented in those cell types:
The 88% mean knockdown of an adipocyte-expressed gene in humans deserves particular emphasis. Adipocytes, fat cells, had been considered extraordinarily difficult to target with nucleic acid therapeutics. They lack the abundant receptor expression that makes hepatocytes so accessible to GalNAc-siRNA. The first ARO-ALK7 clinical data were not just a metabolic disease result; they were a proof point that Arrowhead’s ligand engineering team could identify receptors with sufficient surface expression on almost any cell type and design a TRiM conjugate to exploit them. The same intellectual toolkit is now being applied to neurons and trabecular meshwork cells.
The Leadership of James Hamilton and Tao Pei
The R&D organization that produced this track record operates under the leadership of James Hamilton, M.D., MBA, Chief Medical Officer and Head of R&D, and Tao Pei, Ph.D., Chief Scientific Officer. Hamilton’s involvement in siRNA therapeutics dates to 2005, when he was CEO of multiple RNAi delivery startups before joining Arrowhead. During his time leading clinical development, he oversaw the out-licensing of ARO-HBV (now JNJ-3989) to Janssen Pharmaceuticals, ARO-AAT to Takeda, and ARO-LPA to Amgen, each deal predicated on the scientific quality of Arrowhead’s delivery and knockdown data. Hamilton also led Arrowhead’s in-licensing of Novartis’s RNAi asset portfolio, which enriched the company’s chemical library and broadened the targeting toolkit available to the discovery team.
Tao Pei is the architect of the extrahepatic TRiM delivery platforms that define Arrowhead’s competitive position today. He joined Arrowhead in 2014 as Director of Chemistry, was promoted to Vice President of Discovery Chemistry in 2019, and was appointed Chief Scientific Officer in February 2026, a title that reflects the scope of what he has built. His Ph.D. in Chemistry from Duke University was followed by roles at Merck Process Research and, critically, at Merck Sirna Therapeutics, where he worked specifically on RNAi delivery technologies before joining Arrowhead. That background means Pei arrived with both the medicinal chemistry rigor of a process chemist and the delivery science foundation of someone who had already been working on the hard problem of getting siRNA into cells. He has been solving it at increasing scale ever since.
As Head of Discovery Chemistry before his CSO appointment, Pei built and led multiple functional groups focused on advancing TRiM™ platforms beyond the liver to every major extrahepatic tissue: the central nervous system, skeletal muscle, adipose tissue, heart, eye, and lung. That list is not a description of parallel programs. It is a description of six separate ligand-engineering problems, each requiring identification of a receptor with sufficient expression on the target cell type, design of a targeting moiety with the right binding kinetics, and optimization of the full TRiM conjugate for potency, stability, and tolerability. Pei’s team solved each one. He is also a key architect of TRiM™ itself, and contributed to the final design of Arrowhead’s first TRiM preclinical candidate, olpasiran, now AMG890 at Amgen, advancing in Phase 3. Most directly relevant to this paper: Pei personally presented the ARO-MAPT non-human primate CNS delivery data at the RNA Leaders USA Congress in September 2025, describing potent and long-lasting tau suppression with distribution superior to intrathecal administration. Dr. Agnieszka Glebocka, Vice President of Discovery Chemistry, subsequently presented the full quantitative figures, including the greater than 75% MAPT mRNA knockdown in NHP CNS tissue, at the 7th Annual CNS Delivery Summit on December 10, 2025, formally establishing the NHP benchmark against which the Phase 1/2a human data will be measured.
This author had the privilege of meeting Tao Pei in person at a dinner during a Wisconsin Biohealth Summit, where the conversation turned, naturally, to how Arrowhead chooses which targets to pursue. What emerged from that exchange was something that does not appear in any investor presentation or SEC filing: a picture of how deliberately and rigorously target selection is approached at Arrowhead. Pei’s colleagues at the table were candid in their admiration. The consensus among the Arrowhead scientists present was that Pei brings an unusually refined sense of which targets combine strong genetic validation, tractable biology, and a molecular profile that lends itself to safe, potent knockdown without off-target liability. That is not a small thing. In RNA therapeutics, the choice of target sequence determines whether a drug works at a safe dose or causes unintended silencing of related genes; whether the pharmacodynamic signal is clean or obscured by compensatory biology; whether the platform earns a partner’s trust or loses it in Phase 1. Picking the right target, with the right sequence, against the right gene, in the right tissue, is the foundational act of the entire enterprise. The people who work alongside Pei believe he does this better than almost anyone in the field. That kind of peer recognition, from scientists who see the work up close, is not the sort of evidence that shows up in a discounted cash flow model. But it is exactly the kind of evidence that separates a platform worth owning from one that merely looks like one on paper.
“RNAi is a once-in-a-generation revolution in medicine. Every step forward unlocks the possibility of treating diseases once thought untouchable, and brings real hope to patients still waiting for answers.”
— Tao Pei, Ph.D., Chief Scientific Officer, Arrowhead Pharmaceuticals
The research organization under Hamilton and Pei spans molecular biology, medicinal chemistry, computational biology, pharmacology, and translational medicine across facilities in Pasadena, California, Madison, Wisconsin, and San Diego, California. Pei and his chemistry team are based in Madison, the same city as the Verona manufacturing facility, a geographic proximity that accelerates the feedback loop between discovery synthesis, analytical chemistry, and GMP manufacturing. This distributed but tightly integrated team is what enables Arrowhead to run 19 clinical-stage programs simultaneously, a pipeline breadth that would be operationally impossible without deep bench strength across every function from target identification to IND-enabling package assembly. At most companies, moving a new target from concept to Phase 1 IND filing takes 4-6 years. At Arrowhead, for targets within established tissue types, that timeline can compress to 18-24 months.
The proof is in the cadence. Arrowhead has stated a goal of initiating 2-3 new clinical programs per year. For fiscal year 2026 alone, the company has already dosed first subjects in ARO-MAPT (December 2025) and ARO-DIMER-PA (the world’s first dual-gene siRNA therapeutic). That rate of output from a single discovery organization is not accidental. It reflects a scientific team that has internalized the TRiM design framework so thoroughly that extending it to new tissue types and new molecular targets has become a systematic, repeatable process rather than a heroic one-time achievement.
The multi-tissue delivery record is not just a scientific achievement. It is an investment signal. Every new tissue type Arrowhead has validated has subsequently attracted a large-pharma partnership, generated milestone revenue, or both. Liver delivery: Janssen, Amgen, Takeda, Sarepta deals. Adipose: ongoing. CNS: Novartis, $200M upfront on preclinical data alone. Ocular: next in line. Cardiac: the frontier after that, disclosed at the CNS Delivery Summit and not yet in any model. The scientists are not just doing great science. They are manufacturing strategic optionality that compounds with each new tissue validated.
The Verona Manufacturing Advantage: Speed, Control, and Competitive Moat
A final note on manufacturing that is directly relevant to the BBB platform: the conjugation process that produces the TRiM BBB molecule is not a bespoke, low-yield chemical reaction. The CNS Delivery Summit presentation confirmed that Arrowhead achieves 75–80% total conjugation yield consistently at discovery stage, across scales ranging from milligrams to multi-grams, using equimolar amounts of ligand and siRNA. This is not a laboratory curiosity; it is a manufacturing-grade process. High yield, equimolar stoichiometry, and scale-agnostic consistency are the three properties that make a conjugation chemistry commercially viable. All three are confirmed for the TRiM BBB platform. For investors evaluating whether ARO-MAPT can be manufactured at commercial scale if it succeeds clinically, the answer embedded in these process data is yes.
Behind every successful Phase 1 clinical program is a batch of GMP-grade drug material that had to be manufactured to exacting specifications before a single patient could be dosed. For most small and mid-cap biotechs, this step is a bottleneck that shapes timelines, costs, and competitive position in ways that are rarely discussed but profoundly consequential. The standard practice in the industry is to engage a contract manufacturing organization (CMO), an outside company that manufactures clinical trial material on a fee-for-service basis. Sounds simple. In practice, it is a nine- to eighteen-month wait minimum, with no guarantee of prioritization, no guarantee of first-pass success, and significant transfer of proprietary process knowledge to a third party.
Arrowhead does not operate this way. The company has built its own GMP-grade manufacturing campus in Verona, Wisconsin, a state-of-the-art facility that gives them something most of their peers lack entirely: the ability to manufacture their own clinical trial material on their own timeline, under their own roof, with their own people. The project was first announced in December 2021 and construction broke ground in May 2022. The Verona campus, comprising an approximately 160,000-square-foot GMP manufacturing facility and an approximately 125,000-square-foot laboratory and office building, situated on 13 acres in the Verona Technology Park, was completed in 2024 and represents one of the most significant operational investments in the company’s history.
The speed advantage this confers on Arrowhead’s drug development cycle is difficult to overstate. Consider the arithmetic. A company that must send drug candidate synthesis to an outside CMO waits 9-18 months for a Phase 1 clinical batch. If the formulation is suboptimal, or if preclinical data requires a dosing change, they wait again, and again. Each manufacturing cycle eats 9-18 months and hundreds of thousands to millions of dollars in CMO fees, transfer costs, and team time. Multiply that across 19 clinical programs and a discovery team generating 2-3 new INDs per year, and the delay accumulates into years of lost development time.
At Arrowhead, a manufacturing cycle for a new clinical batch of TRiM-enabled drug can be turned around internally in weeks, not months. Formulation adjustments, dose escalation material, new cohort supply, stability testing batches, all of these can be executed within the Verona facility under the direct oversight of Arrowhead’s own chemistry and manufacturing team. There is no queue. There is no negotiating with a CMO’s scheduling department. There is no knowledge transfer risk. The intellectual property embodied in TRiM manufacturing processes, how to synthesize the targeting ligand, how to conjugate it to the siRNA with the right chemistry and purity, how to formulate it for the intended route of administration, stays inside the building.
This author had the privilege of touring the Verona facility firsthand, and what the visit makes clear is how much flexibility the in-house operation provides that outside contractors simply cannot. Arrowhead can adjust formulations, change doses, add cohorts, and respond to emerging preclinical data on its own schedule, without negotiating slots, without the knowledge transfer costs of re-engaging a CMO, and without ceding process control over the most sensitive intellectual property in the company. This is a meaningful structural advantage over virtually every competitor in the RNAi space, with the notable exception of Alnylam, which has invested in its own manufacturing infrastructure as well. Every other company working in RNA therapeutics largely depends on external manufacturing networks that introduce delay, cost, and coordination overhead at every development milestone. Arrowhead has removed those dependencies across a 19-program pipeline, and the compounding time savings that result over the course of a drug’s development from IND to NDA are substantial.
CEO Christopher Anzalone said plainly about the facility during the Q1 2022 earnings call: “We view this as a strong competitive advantage.” That is an understatement. In a field where the speed of clinical iteration determines who gets to pivotal data first, Arrowhead has structurally removed one of the industry’s most common rate-limiting steps. When a preclinical result in the CNS ocular program requires a modified molecule, the Verona team can manufacture it. When ARO-MAPT dose escalation proceeds faster than expected, the supply can keep pace. When a partner like Novartis requests GMP batches for a preclinical CTA package, Arrowhead can deliver without outsourcing the most sensitive part of the process.
The Verona facility also positions Arrowhead uniquely for commercial-scale manufacturing. The GMP building was designed from the ground up to scale from clinical trial quantities to commercial launch quantities for TRiM-enabled therapeutics. For a company with Redemplo already on the market and multiple programs approaching potential NDA/BLA filings, the ability to grow into commercial manufacturing without a facility build or another CMO transition is a meaningful cost-and-timeline advantage that will compound as the pipeline matures.
Most biotechs wait 9-18 months just to get a Phase 1 drug batch made. Arrowhead makes their own, in their own facility, on their own schedule, with the flexibility to respond to data in real time without the delays and costs that come with external manufacturing. Outside of Alnylam, no major RNAi company operates at this level of manufacturing self-sufficiency. Across a 19-program pipeline, that advantage compounds into years of recovered development time and a meaningfully faster path to data, approval, and commercial launch.
XIV. Risk Factors
This analysis presents a bullish case for ARO-MAPT and ARWR. But intellectual honesty requires a rigorous examination of the risks. The following represent the primary concerns an investor should hold in mind.
Clinical and Scientific Risks
BBB Penetration May Not Translate from NHP to Human: Non-human primate models are the best available preclinical predictor of CNS biodistribution, but translation to humans is not guaranteed. Arrowhead has disclosed greater than 75% MAPT mRNA knockdown in NHP CNS tissue at clinically translatable doses, which is a strong preclinical foundation and directly comparable to the NHP data that informed BIIB080’s successful Phase 1. However, the human BBB has structural differences from NHP. If human PK data shows insufficient CNS exposure after subcutaneous injection, or if the knockdown depth observed in NHPs does not replicate in humans, the platform thesis is materially impaired. This remains the single most important risk in the entire investment case.
Off-Target CNS Effects: Delivering a gene-silencing molecule broadly across the CNS carries inherent risk. Any off-target knockdown of neurologically important genes could produce adverse neurological events. The safety review in Phase 1 will be scrutinized carefully.
Deep Tau Reduction and Physiological Function: Tau has established physiological roles in axonal transport and microtubule stability. Preclinical models suggest that very deep tau suppression, above 80–85% sustained over long periods, may produce subtle effects in some systems. Arrowhead’s trial design incorporates NfL monitoring specifically to detect axonal injury. The dosing regimen, which achieves 70–80% mRNA knockdown rather than near-complete ablation, appears to land in a range that is biologically active without approaching the suppression levels associated with adverse consequences in animal models. BIIB080’s Phase 1b provided human evidence that this range is safe: no dose-limiting neurological events were observed at doses achieving 56–60% CSF tau reduction. Nevertheless, this risk deserves explicit acknowledgment and will be a primary focus of the Phase 1 safety review.
Tau Biology Complexity: Partial tau reduction might not be sufficient to arrest aggregation once disease pathology is established. Even deep tau knockdown might not slow cognitive decline if other pathological processes (neuroinflammation, synaptic loss) have already progressed significantly.
Isoform Selectivity Considerations: All six tau isoforms are targeted by ARO-MAPT. While this is the goal, the 4-repeat and 3-repeat isoform balance is physiologically important. Disproportionate reduction of one isoform could theoretically produce unintended consequences.
Competitive Risks
BIIB080 Efficacy Data Could Define the Field: If the BIIB080 Phase 2 CELIA trial reports strong efficacy data in 2026, Biogen and Ionis would become the standard-of-care benchmark. This would not eliminate the market for a subcutaneous alternative, but it would shift the regulatory burden of proof for ARO-MAPT and change the partnership dynamic.
Alnylam’s CNS Ambitions: Alnylam (ALNY) has announced development of intrathecal siRNA approaches and is the dominant force in global RNA therapeutics. A pivot by Alnylam toward subcutaneous CNS delivery would introduce the most credible competitive threat to ARO-MAPT’s differentiation.
Regulatory Risks
CNS RNAi Is Uncharted Regulatory Territory: No RNAi therapeutic delivered subcutaneously to the CNS has received FDA approval. The regulatory framework for this modality is still being defined. IND-enabling requirements, safety monitoring, and approval standards could evolve in ways that add time and cost.
ARIA-like Concerns: The FDA required extensive ARIA monitoring for anti-amyloid antibodies. Whether similar monitoring will be required for tau-reducing RNAi therapeutics is unknown, but any neuroimaging requirement adds complexity and cost to the clinical program.
Execution Risks
Enrollment Timeline Risk: The AD patient cohort (up to 48 subjects with early AD) requires biomarker-confirmed diagnosis and may take longer to enroll than projected. Delays in AD patient data could extend the timeline to pivotal data readouts.
Capital Deployment: The convertible notes offering in January 2026 and concurrent equity offering diluted shareholders and added $350M+ in debt. While management deployed this capital toward program advancement, the burden of demonstrating value from that capital is real.
XV. Conclusion: The CNS Frontier Opens
Arrowhead Pharmaceuticals has spent a decade building one of the most productive RNA therapeutics platforms in the industry. It has commercial-stage products, near-commercial assets, multiple deep-pocket partnerships, and a balance sheet that no longer constrains its ambitions, and yet, despite all of this, the most significant thing Arrowhead may do in 2026 is small enough to fit in a subcutaneous syringe.
ARO-MAPT is not a guaranteed success. No clinical program is. The blood-brain barrier remains the most formidable challenge in drug delivery, and the history of Alzheimer’s drug development is littered with programs that worked in animals and failed in humans. Intellectual humility requires acknowledging this possibility.
But here is what we know:
The tau hypothesis is among the most genetically validated targets in neurodegenerative disease, and the genetic case for MAPT silencing specifically is airtight.
BIIB080’s clinical data have already proven that tau reduction is pharmacologically achievable and safe in humans. The mechanism works. The question is whether ARO-MAPT can do it better.
Arrowhead’s NHP data show 70–80% MAPT mRNA knockdown (up to 85% cortex) and 50–60% CSF tau protein reduction via subcutaneous dosing, with distribution across 14 measured brain regions ranging only 3x from highest to lowest. The NHP CSF tau result is already in the range of BIIB080’s best human Phase 1b data. No other company has presented comparable subcutaneous CNS delivery data in primates.
Novartis paid $200 million upfront for a preclinical CNS RNAi program on this same delivery platform. That is the disclosed floor of what the technology is worth before human data exists.
Arrowhead’s indication ladder strategy, PSP and CBD before Alzheimer’s, FTD-MAPT before sporadic disease, smaller before larger, means value creation is staged, de-risked, and begins well before any Alzheimer’s Phase 3 trial reads out.
The tauopathy market spans multiple indications, multiple orphan regulatory pathways, multiple partnership opportunities, and one overarching theme: there are no approved disease-modifying treatments for any of them. Arrowhead is first through the door.
Arrowhead is the only company with a clinical-stage subcutaneous CNS RNAi program currently enrolling patients. That is not a minor distinction. It is the definition of competitive advantage in drug delivery.
Every other CNS gene silencing program in existence, including all five of Alnylam’s CNS programs disclosed in their 2025 Annual Report, uses intrathecal or IV delivery. Alnylam has not disclosed a subcutaneous BBB delivery solution. Neither has Ionis, Lilly, Biogen, or any other major RNAi or ASO company. Arrowhead is not competing with these programs on equivalent terms. If subcutaneous delivery is validated, it is structurally advantaged over every intrathecal program that a physician, patient, partner, or payor must choose between.
If ARO-MAPT achieves meaningful tau knockdown via subcutaneous administration in humans, the implications cascade across all layers of the investment thesis simultaneously: the drug works, the platform is validated, the indication ladder is activated, the partnership economics improve dramatically, and the acquisition premium expands. These are not independent events. They are the same event viewed from different angles, and each angle adds value that compounds on the others.
The market is currently pricing Arrowhead as a cardiovascular and metabolic RNA therapeutics company with an interesting but unproven CNS program. This is precisely the mismatch that creates opportunity. History in the RNA therapeutics space, and in Arrowhead’s own history specifically, is consistent on one point: platform inflection points are almost never priced in advance. The first successful GalNAc-siRNA liver data at Alnylam. Sarepta’s first DMD exon-skipping results. Arrowhead’s own early knockdown data that preceded years of subsequent re-rating. The pattern is clear. The markets wait. The data arrives. The valuation catches up.
The 2H 2026 ARO-MAPT data readout is that moment in waiting, and for investors who understand the indication ladder, the CNS delivery breakthrough, and the platform implications that extend far beyond a single tau program, the case for accumulating ARWR ahead of that readout has rarely been clearer. Arrowhead does not need to cure Alzheimer’s in 2026 to be worth substantially more than it is today. It simply needs to show that its new CNS delivery system works in humans. The rest, the orphan indications, the precision medicine ladder, the Alzheimer’s Phase 3, the partnerships, the acquisitions, follows from that single event.
The year of tau has arrived. The CNS frontier is open. The indication ladder is in place. Arrowhead enters 2H 2026 with NHP data showing 70–80% MAPT mRNA knockdown across every measured brain region, up to 85% in cortex, and 50–60% CSF tau protein reduction sustained for 5 months, both via subcutaneous injection. The NHP CSF tau data already occupies the same therapeutic range as BIIB080’s best human Phase 1b result. What remains is the human translation step, and Arrowhead is the only company at the base of the tauopathy ladder with a subcutaneous key already in human hands. Alnylam, Ionis, Biogen, Lilly, and every other company that has committed to intrathecal CNS gene silencing built those programs on the assumption that subcutaneous CNS delivery was not possible. The 2H 2026 data will test that assumption directly. If Arrowhead proves it wrong, the competitive landscape of CNS drug delivery does not gradually shift, it breaks.
Glossary of Key Terms
The following definitions are provided for readers who may be less familiar with the clinical, scientific, and financial terminology used throughout this paper. Terms are listed alphabetically.
Accelerated Approval
An FDA pathway allowing drug approval based on a surrogate endpoint reasonably likely to predict clinical benefit, with a confirmatory trial required post-approval. Applicable to PSP and CBD given the availability of validated tau biomarker surrogates and the absence of any approved disease-modifying therapy.
Amyloid-Beta (Aβ)
A protein fragment that accumulates as extracellular plaques in Alzheimer’s disease brains. The primary target of lecanemab (Leqembi) and donanemab (Kisunla). Amyloid pathology precedes tau pathology temporally, but tau density correlates more tightly with cognitive decline and neurodegeneration.
ALN-5288
Alnylam Pharmaceuticals’ investigational siRNA targeting MAPT mRNA for Alzheimer’s disease, disclosed in the Alnylam 2025 Annual Report (released April 7, 2026) as part of their Alnylam 2030 five-year strategic plan. Delivered intrathecally. Pharmacologically equivalent to ARO-MAPT in mechanism: both silence MAPT to reduce tau protein production. Critically, the Alnylam 2030 plan, which represents a five-year board-level strategic commitment, contains no mention of subcutaneous CNS delivery of any kind. If ARO-MAPT validates subcutaneous BBB crossing in humans, ALN-5288 becomes the inferior-modality version of the same drug, and Alnylam’s entire five-year CNS strategy faces reassessment.
ARIA (Amyloid-Related Imaging Abnormalities)
Brain imaging abnormalities, including edema (ARIA-E) and microhemorrhages (ARIA-H), occurring as side effects of anti-amyloid antibody therapies. Require MRI monitoring and complicate administration of lecanemab and donanemab. Not applicable to tau-targeting approaches such as ARO-MAPT.
ARO-MAPT
Arrowhead Pharmaceuticals’ investigational siRNA therapeutic targeting the MAPT gene to reduce tau protein production in CNS cells. Currently in Phase 1/2a clinical trial AROMAPT-SC-1001 (NCT07221344). Delivered subcutaneously using the TRiM BBB platform.
ASO (Antisense Oligonucleotide)
A synthetic single-stranded DNA-like molecule that binds target mRNA and recruits RNase H for degradation, reducing protein production. The modality of BIIB080. ASOs and siRNA both reduce mRNA but differ in mechanism: ASOs require stoichiometric hybridization; siRNA operates through catalytic RISC-mediated cleavage.
BBB (Blood-Brain Barrier)
A highly selective semipermeable border of endothelial cells separating circulating blood from brain extracellular fluid. Prevents most large molecules, including naked siRNA and ASOs, from reaching the brain after systemic administration. Crossing the BBB is the central delivery challenge in CNS therapeutics and the primary innovation of the TRiM BBB platform.
BIIB080 (ION464 / MAPTRx / Diranersen)
The Ionis/Biogen antisense oligonucleotide targeting MAPT mRNA, delivered intrathecally. Achieved 56–60% CSF total-tau reduction in Phase 1b. Currently in Phase 2 CELIA trial (NCT05399888, 416 patients). The primary clinical benchmark for ARO-MAPT.
Biomarker
A measurable biological indicator used to assess disease status, drug activity, or treatment response. Key biomarkers in this paper: CSF total-tau, CSF p-tau181/217, plasma p-tau217, NfL, and tau PET.
Breakthrough Therapy Designation (BTD)
An FDA designation expediting development and review of drugs for serious conditions where preliminary clinical evidence indicates substantial improvement over available therapy. Provides intensive FDA guidance and rolling review. Applicable to PSP and CBD as potential ARO-MAPT indications given the complete absence of any approved disease-modifying therapy.
CBD (Corticobasal Degeneration)
A rare progressive 4-repeat tauopathy with tau aggregates in both neurons and glial cells (astrocytes and oligodendrocytes) in cortical and subcortical regions. No approved disease-modifying therapy. First-rung indication on the ARO-MAPT tauopathy ladder alongside PSP.
Cardiomyocyte
The primary contractile muscle cell of the heart. Post-mitotic, post-natal cells that represent one of the most technically challenging targets for nucleic acid therapeutics due to limited endocytic activity, dense extracellular matrix, and rapid cardiovascular clearance of macromolecules. Arrowhead disclosed a preclinical TRiM platform for cardiomyocyte delivery at the 7th Annual CNS Delivery Summit (December 2025). Key potential targets: PLN (phospholamban) for heart failure, MYH7/MYBPC3 for hypertrophic cardiomyopathy, and TTR for ATTR-CM.
CELIA Trial
Biogen and Ionis’s Phase 2 clinical trial of BIIB080 (NCT05399888) in early Alzheimer’s disease. 416 patients enrolled as of April 2025. Primary endpoint: change from baseline in CDR-SB. Data expected 2026. The most important comparative readout in the tau field.
CNS (Central Nervous System)
The brain and spinal cord. The target tissue for ARO-MAPT and the anatomical site of tau pathology across tauopathies.
Cryo-EM (Cryogenic Electron Microscopy)
An imaging technique resolving molecular structures at near-atomic precision by flash-freezing and electron-beam imaging. Used by Arrowhead to confirm the TRiM BBB ligand binds TfR1’s apical domain without displacing endogenous transferrin, establishing the mechanistic and safety foundation for the BBB platform.
CSF (Cerebrospinal Fluid)
The clear fluid surrounding and cushioning the brain and spinal cord. The primary sampling matrix in neurological drug development. Tau protein concentrations in CSF, measured by lumbar puncture, are the key pharmacodynamic biomarker for tau-targeting therapies.
CTA (Clinical Trial Authorization)
Regulatory clearance to initiate a clinical trial in countries outside the United States. The equivalent of the FDA’s IND. Arrowhead filed a CTA on September 10, 2025 to initiate the ARO-MAPT Phase 1/2a trial.
CTE (Chronic Traumatic Encephalopathy)
A progressive tau-driven neurodegenerative disease caused by repetitive head trauma. Affects millions of current and former contact sport athletes, military veterans, and others with repeated head impact histories. No approved therapy. A long-horizon indication on the ARO-MAPT indication ladder.
Deep Brain Structures
Brain regions distant from the CSF-contacting ventricular and subarachnoid surfaces, including the substantia nigra, subthalamic nucleus, globus pallidus, red nucleus, and brainstem nuclei. Primary sites of tau pathology in PSP. Structurally under-dosed by intrathecal delivery due to CSF concentration gradients; fully accessible to vascular delivery via TfR1.
DIAN-TU
Dominantly Inherited Alzheimer Network Trials Unit. The academic consortium that established FDA precedent for presymptomatic trials in familial Alzheimer’s disease using biomarker endpoints. Directly relevant to the FTD-MAPT prevention trial design.
EOP2 Meeting (End-of-Phase 2)
A formal Type B FDA meeting in which a sponsor presents the Phase 2 dataset and seeks guidance on Phase 3 design, endpoints, and registration pathway. For ARO-MAPT, expected mid-2027, this meeting will determine whether a dedicated AD Phase 2b is required before Phase 3, or whether an adaptive seamless design is acceptable.
FCS (Familial Chylomicronemia Syndrome)
An ultra-rare genetic disorder caused by LPL loss-of-function mutations, producing severely elevated triglycerides and recurrent acute pancreatitis. The first approved indication for plozasiran (Redemplo). Arrowhead’s proof-of-concept for the orphan-first indication ladder strategy applied to ARO-MAPT.
FTD (Frontotemporal Dementia)
The second most common dementia in adults under 65. A subset is caused by dominant MAPT mutations (FTD-MAPT), enabling a presymptomatic prevention trial design in genetically identified at-risk individuals before symptom onset.
GalNAc (N-Acetylgalactosamine)
A sugar molecule used as a targeting ligand in liver-directed RNAi therapeutics. Binds the ASGPR receptor on hepatocytes with high affinity. The foundation of Arrowhead’s liver TRiM platform and the delivery mechanism behind plozasiran. GalNAc does not cross the blood-brain barrier, which is why a different targeting ligand (TfR1) was required for the CNS platform.
GMP (Good Manufacturing Practice)
The regulatory standard governing pharmaceutical manufacturing, ensuring consistency, safety, and quality. Arrowhead’s Verona, Wisconsin facility operates three parallel GMP production lines capable of supporting nine programs across multiple development stages.
IHC (Immunohistochemistry)
A laboratory technique using antibodies to detect specific proteins or nucleic acids in tissue sections. RNAscope multiplex IHC, used in the CNS Delivery Summit dataset, simultaneously identifies MAPT mRNA and labels neurons, astrocytes, microglia, and oligodendrocytes to confirm pan-cellular knockdown in NHP brainstem.
IND (Investigational New Drug Application)
The application filed with the FDA to obtain permission to conduct clinical trials in the United States. Required before initiating any US clinical trial.
Indication Ladder
Arrowhead’s development strategy of initiating parallel trials in smaller, faster, orphan-eligible disease populations (PSP, CBD, FTD-MAPT) before or alongside the larger Alzheimer’s program, generating earlier approvals, real-world safety data, and commercial revenue that de-risk and accelerate the Alzheimer’s path. Modeled on the plozasiran template: FCS approval first, sHTG second.
Intrathecal (IT) Delivery
Administration of a drug via lumbar puncture directly into the cerebrospinal fluid. Used by BIIB080 and all other current CNS ASO and siRNA programs except ARO-MAPT and AL064. Creates a rostro-caudal concentration gradient that systematically under-doses deep brain structures distant from the injection site.
IP (Intellectual Property)
Patents, trade secrets, and proprietary know-how protecting Arrowhead’s TRiM BBB delivery platform, including the TfR1-targeting small-molecule ligand architecture and the covalent conjugation chemistry, creating a barrier to competitive replication.
MAPT
Microtubule-Associated Protein Tau. The gene on chromosome 17 encoding all six tau isoforms in the adult brain. Dominant mutations cause familial FTD and PSP. The gene targeted by ARO-MAPT and all other tau-silencing therapeutics in development.
MCI (Mild Cognitive Impairment)
A clinical stage between normal aging and dementia: cognitive changes noticeable to the patient and confirmed by testing, but not yet severely impairing daily function. MCI due to AD is the earliest eligible disease stage in ARO-MAPT’s Phase 1/2a AD cohort.
mRNA (Messenger RNA)
The molecular intermediary between a gene’s DNA blueprint and the protein it encodes. Both ARO-MAPT (siRNA) and BIIB080 (ASO) target and degrade MAPT mRNA to prevent tau protein production before it is translated.
NFT (Neurofibrillary Tangle)
An intraneuronal accumulation of hyperphosphorylated tau protein in paired helical filament structures. A hallmark neuropathological feature of Alzheimer’s and other tauopathies. NFT density correlates more strongly with cognitive decline than amyloid plaque burden.
NfL (Neurofilament Light Chain)
A structural protein released from damaged axons into CSF and blood. A biomarker of neurodegeneration and axonal injury. Monitored in ARO-MAPT’s Phase 1/2a to confirm that tau reduction is not accompanied by neuronal damage.
NHP (Non-Human Primate)
Cynomolgus macaques (Macaca fascicularis) used in preclinical ARO-MAPT and BIIB080 CNS delivery and knockdown studies. The closest available animal model to human BBB physiology. TfR1 is confirmed cross-reactive between cynomolgus macaques and humans, the molecular basis for expecting human BBB penetration to mirror NHP results.
Oligodendrocyte
A glial cell responsible for myelinating axons in the CNS. Tau inclusions in oligodendrocytes (coiled bodies) are a hallmark of PSP and CBD pathology. ARO-MAPT’s confirmed pan-cellular knockdown, including oligodendrocytes, is clinically relevant to these indications in a way that BIIB080’s intrathecal delivery profile may not be.
Orphan Drug Designation (ODD)
An FDA designation for drugs treating diseases affecting fewer than 200,000 Americans. Confers seven years of market exclusivity from approval, priority review eligibility, FDA fee waivers, and tax credits for qualifying trials. PSP, CBD, and FTD-MAPT all qualify. The regulatory foundation of Arrowhead’s indication ladder.
p-tau (Phospho-Tau)
Hyperphosphorylated forms of tau protein, including p-tau181 and p-tau217, elevated in CSF and plasma of AD and tauopathy patients. More directly linked to pathological aggregation than total-tau. Increasingly measurable from blood rather than CSF, enabling non-invasive monitoring.
PDUFA Date
Prescription Drug User Fee Act date: the FDA’s target review completion date for an NDA or BLA. Typically 12 months after filing (standard review) or 6 months (priority review).
PD (Pharmacodynamics)
The study of a drug’s biological effects on the body. In this paper: CSF tau reduction, plasma p-tau217 changes, and NfL levels are the primary pharmacodynamic endpoints for ARO-MAPT.
PK (Pharmacokinetics)
The study of how the body processes a drug: absorption, distribution, metabolism, and excretion. PK data from AROMAPT-SC-1001 will characterize CNS tissue exposure after subcutaneous dosing, establishing the concentration-effect relationship that the NHP PK/PD model projects.
PSP (Progressive Supranuclear Palsy)
A rapidly progressive, uniformly fatal 4-repeat tauopathy defined by tau accumulation in the subthalamic nucleus, substantia nigra, globus pallidus, and brainstem nuclei. Clinical hallmarks: vertical gaze palsy, postural instability, dysarthria. No approved disease-modifying therapy. The highest-priority orphan indication on the ARO-MAPT indication ladder.
Receptor-Mediated Transcytosis
The mechanism by which the TRiM BBB conjugate crosses brain endothelial cells: binding TfR1 on the luminal (blood) surface, internalization, vesicular transport across the cell, and release into brain parenchyma on the abluminal side. Enables systemic (subcutaneous) delivery to achieve brain exposure without lumbar puncture.
RISC (RNA-Induced Silencing Complex)
The intracellular molecular machinery executing RNAi-mediated gene silencing. The antisense strand of the siRNA is loaded into RISC, which guides sequence-specific mRNA cleavage. RISC is catalytic: one loaded complex cleaves multiple mRNA molecules, enabling deep and durable knockdown from infrequent dosing.
RNAi (RNA Interference)
A natural cellular gene-silencing mechanism triggered by double-stranded RNA, inducing sequence-specific mRNA degradation. Arrowhead’s TRiM platform harnesses this pathway by delivering synthetic siRNA duplexes to target cells. The mechanistic basis of every Arrowhead therapeutic program.
rNPV (Risk-Adjusted Net Present Value)
The standard biotech analyst valuation methodology. Applies probability-of-success estimates to projected future cash flows, discounted to present value. As this paper argues, rNPV systematically undervalues platform-enabling programs like ARO-MAPT by pricing the drug asset but assigning near-zero value to the delivery technology validation it represents.
RNAscope
A proprietary in situ hybridization technique enabling visualization and quantification of specific mRNA molecules at single-cell resolution in tissue sections. Used in the CNS Delivery Summit dataset to confirm MAPT mRNA knockdown across all four major CNS cell types in NHP brainstem.
sHTG (Severe Hypertriglyceridemia)
Fasting triglycerides above 500 mg/dL; associated with elevated acute pancreatitis risk. The second major plozasiran indication, currently in Phase 3 SHASTA-3 and SHASTA-4 trials with data expected 2H 2026.
siRNA (Small Interfering RNA)
A 21–23 nucleotide double-stranded RNA molecule directing RISC-mediated sequence-specific mRNA cleavage. The molecular payload of ARO-MAPT. Mechanistically distinct from ASOs: siRNA uses catalytic RISC cleavage; ASOs use stoichiometric RNase H recruitment.
SRP-1005
Sarepta Therapeutics’ investigational CNS RNAi program targeting HTT (huntingtin) for Huntington’s disease, using Arrowhead’s TRiM BBB delivery platform. CTA filed Q1 2026. Licensed from Arrowhead as ARO-HTT. Arrowhead has also licensed ARO-ATXN2 (targeting ataxin-2 for SCA2 and ALS) to Sarepta on the same platform. Together, SRP-1005 and ARO-ATXN2 represent two independent Sarepta commitments to the TRiM BBB platform, alongside Novartis’s ARO-SNCA, for a total of three large-pharma CNS program licenses on a single delivery technology.
Substantia Nigra
A midbrain structure critical to motor control. Degeneration of the substantia nigra pars reticulata is a defining feature of PSP. Measured at 0.47 μg/g drug concentration in ARO-MAPT NHP distribution data, producing 70–80% MAPT mRNA knockdown despite being the lowest-concentration region of the 14 measured.
Tau
A microtubule-associated protein encoded by MAPT. In healthy neurons: stabilizes microtubules, facilitates axonal transport. In tauopathies: becomes hyperphosphorylated, detaches from microtubules, and aggregates into neurofibrillary tangles. The target protein for ARO-MAPT, BIIB080, and all tau-silencing programs in development.
Tau PET
Positron emission tomography imaging using radioactive tracers (e.g., flortaucipir) that bind tau aggregates, providing a spatial map of tau pathology in the living brain. An exploratory endpoint in ARO-MAPT’s Phase 1/2a and a surrogate endpoint candidate for accelerated approval in PSP/CBD.
Tauopathy
Any neurodegenerative disease defined by pathological tau protein accumulation. Encompasses Alzheimer’s disease, PSP, CBD, FTD-MAPT, CTE, and Pick’s disease. Collectively affects more than 40 million people globally. ARO-MAPT’s pan-CNS distribution, including deep brain structures, makes it pharmacologically relevant to every tauopathy.
TfR1 (Transferrin Receptor 1)
A cell surface receptor with high expression on brain endothelial cells due to the brain’s iron demand. The molecular target of Arrowhead’s TRiM BBB ligand for receptor-mediated transcytosis. The TRiM BBB ligand binds TfR1’s apical domain without displacing endogenous transferrin, preserving normal iron transport.
TRiM Platform (Targeted RNAi Molecule)
Arrowhead’s proprietary modular drug design framework combining a siRNA duplex with a cell-type-specific targeting ligand through optimized linker chemistry. Validated across liver, lung, skeletal muscle, adipose, CNS, and other tissue types. ARO-MAPT uses the TRiM BBB variant incorporating the TfR1-targeting ligand.
TRiM BBB Platform
The CNS-specific TRiM variant using a small-molecule TfR1-targeting ligand attached to siRNA through a stable non-reversible covalent linkage. Achieves receptor-mediated BBB transcytosis after subcutaneous dosing. First human dosing: December 2025 (ARO-MAPT). Cross-reactive between cynomolgus macaques and humans.
WAC (Wholesale Acquisition Cost)
A drug’s list price before discounts, rebates, or negotiated reductions. Net realized price is typically 20–40% lower. Used in this paper when estimating ARO-MAPT revenue potential.
A Note on Supporting Independent Research
These white papers took hundreds of hours to produce. The asset inventory, valuation methodology, bidder analysis, comparable transaction work, acquisition thesis, competitor analysis, supporting charts, and science analytics are the result of deep primary research and is not available in sell-side coverage. Most of the analysis presented represents independent research not published elsewhere. It is being shared freely because the thesis deserves the widest possible audience. Every Arrowhead shareholder benefits from a well-informed market that understands what the data means and what the asset is worth. That is why this paper exists.
If this research has been valuable to you, whether it shaped your thinking, validated your conviction, or simply saved you the time of doing this work yourself, a voluntary contribution is genuinely appreciated and directly funds the next paper.
For individual investors and readers
Any amount you feel reflects the value you received is welcome and meaningful. A contribution in the range of what you might pay for a single premium research report is a thoughtful gesture that makes a real difference.
For family offices, investment funds, hedge funds, and research platforms
This paper is the caliber of work that institutional research desks bill significant retainers to produce. If your team referenced it, distributed it internally, or used it to inform a position, a suggested contribution of $1,500 or more reflects the professional value of the analysis, though any amount is meaningful. Your support makes it possible to continue publishing at this level without a paywall that limits the reach of the ideas. If your organization requires an invoice to process a payment, please reach out directly at bioboyscout@gmail.com and one will be provided promptly.
There is no obligation and no expectation. This is purely a thank you for work that meant something to you.
Zelle: (847) 227-7909
Thank you for reading, and for being part of a community that takes this thesis seriously.
— Robert Toczycki | BioBoyScout
Important Risks, Disclosures, & Disclaimers
The author, Robert Toczycki (aka BioBoyScout), certifies that:
· all views expressed in this white paper accurately reflect his personal opinions about the topic discussed; and
· he was not compensated in any form for producing this white paper.
The author currently holds a position in Arrowhead Pharmaceuticals, Inc.
This white paper is prepared for informational purposes only and is intended for qualified institutional investors and sophisticated market participants. It does not constitute investment advice, a solicitation, or an offer to buy or sell any security. All opinions expressed herein represent the views of BioBoyScout at the time of publication and are subject to change without notice. Past performance of any drug modality, device category, or company discussed herein is not indicative of future results. Investing in biotech and life sciences companies involves substantial risk, including the risk of complete loss of invested capital. All clinical data, market projections, and financial estimates referenced in this document are sourced from publicly available information and are subject to revision as new information becomes available. This document has not been reviewed, approved, or endorsed by any regulatory authority. Recipients should conduct their own due diligence and consult with their own financial, legal, and regulatory advisors before making investment decisions based on the information contained herein.
About the Author
Robert Toczycki is an independent analyst with a JD and MBA, as well as a BS in Mathematics and Computer Science from the University of Illinois in Champaign. He has a deep passion for financial analysis, particularly identifying valuation discrepancies and demonstrating them through rigorous, data-driven research and solid analytics.
Comments or questions: bioboyscout@gmail.com.
Copyright © 2026, BioBoyScout. All Rights Reserved.